April 10, 2023 at 3:39 pm | Updated April 15, 2026 at 3:41 am | 10 min read
Photosynthesis is one of the most important physiological processes for plants and ecosystems. The number of areas and themes are increasing as research on this topic gets multidisciplinary due to its potential applications. Measurement and analysis of photosynthesis are necessary for these studies. This blog covers the top 10 questions plant scientists consider about photosynthesis before choosing analysis methods and tools.
1. What are the different types of photosynthesis, and how do they differ?
There are three types of photosynthesis C3, C4, and CAM, depending on where and when the two steps of photosynthesis occur. The first step is the light-dependent stage when light is absorbed and converted to chemical energy; the light splits water molecules and fixes carbon dioxide (CO2) to give ATP and NADPH. In the second light-independent stage, or Calvin-Benson Cycle, the chemical energy stored in ATP and NADPH produces sugar or glucose.
In C3 photosynthesis, the first carbon compound has three carbon atoms. The two stages of photosynthesis occur in the same chloroplast, which is situated in the mesophyll cells. The enzyme, Rubisco, which fixes CO2, can also react with oxygen (O2). In C3 photosynthesis, 20 percent of the Rubisco is involved in fixing O2 in photorespiration, which reduces photosynthetic efficiency; see Figure 1.
Subscribe to receive our monthly round-up of articles.
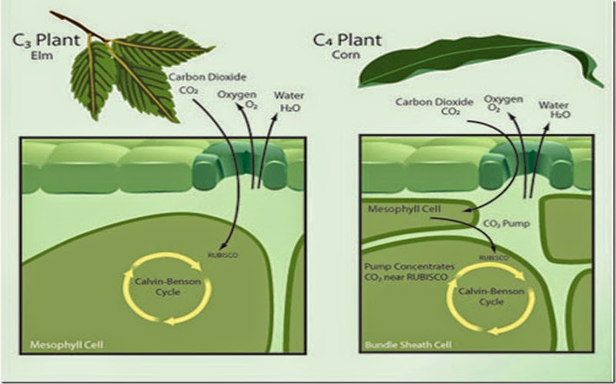
Figure 1: Difference between C3 and C4 photosynthesis, Meacham-Hensold, 2020. (Image credits: https://ripe.illinois.edu/blog/difference-between-c3-and-c4-plants)
C4 photosynthesis produces 4-carbon compounds as the initial product of photosynthesis. C4 photosynthesis occurs in two different parts of a leaf to avoid photorespiration. CO2 fixation occurs in mesophyll cells using PEP carboxylase, insensitive to O2. Since there is little or no Rubisco in the mesophyll of C4 plants, there is no photorespiration. The fixed CO2 is then sent to the bundle sheath cells, where Rubisco is concentrated, and the Calvin-Benson Cycle occurs; see Figure 1. In some cases, the two stages of photosynthesis do not occur in two different cells but just in two separate portions of a cell.
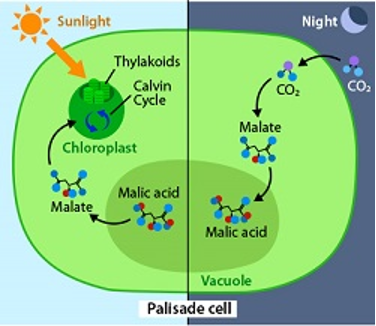
Figure 2: “In CAM plants, carbon dioxide is only gathered at night, when the stomata open.” The rest of photosynthesis happens during the day in the presence of light, Kropp and Halasey, 2014. (Image credits: https://askabiologist.asu.edu/cam-plants)
CAM is a special kind of C4 photosynthesis, where there is a temporal separation of the two stages in a single cell.
CAM occurs in xeric conditions, where the stomata are closed daily to prevent water vapor loss. Therefore, the leaves take CO2 at night and convert it to malic acid. During the day, the malic acid is converted to CO2, and the two photosynthesis stages occur in light. This photosynthesis is called Crassulacean Acid Metabolism (CAM) because carbon dioxide is stored as an acid at night.
2. How can photosynthesis be measured?
There are many ways in which photosynthesis can be measured. The standard methods are discussed below:
Gas exchange: Photosynthesis is a chemical reaction, and estimating changes in the initial compounds and final products is the standard way of measuring the process. Gas exchange analyzers measure the amount of carbon dioxide fixed in photosynthesis and oxygen produced by estimating levels of the two gases at the inlet and outlet, i.e., before and after a leaf conducts photosynthesis.
These types of measurements are possible in closed or open systems. In open systems, the incoming air is drawn from the ambient atmosphere or passed through control modules, and oxygen is released into the air. In closed systems, the air after photosynthesis is again circulated back to the inlet. Natural gas exchange estimations are possible onsite in farms, forests, greenhouses, and the laboratory and are non-destructive.
Chlorophyll fluorescence: Another non-destructive method is estimating the light not used in photosynthesis, which is reflected as red and far-red wavelengths called fluorescence radiation. The fluorescence radiation is complementary to the rate of photosynthesis. It estimates photosynthetic efficiency in the field through measurement by portable tools or is captured by large-scale remotely-sensed multispectral or hyperspectral images.
Hill’s reaction: In photosynthesis, the water molecules are split to produce O2 and hydrogen or electron. Hill’s reaction transfers the electron to an electron acceptor (NADP+). A dye is used as an artificial electron acceptor to measure Hill’s reaction. The dye DCIP (2,6-dichlorophenolindophenol) changes from blue to colorless when the electron reduces it and helps estimate the light-dependent phases of photosynthesis.
Indirect means of estimating photosynthesis involve measuring dry matter and biomass accumulation due to the amount of photosynthesis conducted by a plant.
3. How do plants optimize photosynthesis under other environmental conditions (e.g., light, temperature, CO2 concentrations)?
As light intensity increases, the amount of photosynthesis will increase. However, too much light will limit photosynthesis, cause sunburn, and even cause cell death. Most plants use photo-protective thermal dissipation to remove nearly half the light that falls on them. In the case of slow-growing evergreen, this can be as high as 90-95 percent. Increasing chlorophyll fluorescence is another possibility.
Plants also make the angles of leaves steeper to avoid too much light absorption. And move or synthesize more chloroplasts on the leaf top to absorb light and conduct photosynthesis. Conversely, some plants have low chlorophyll in sunny sites. In arid regions, plants will also reflect light by growing hairs and a thicker protective wax layer on leaves to avoid decreased photosynthesis.
As CO2 levels increase, so will the photosynthetic efficiency. The current CO2 level is ≈ 0.038%, and plants are expected to increase photosynthesis and absorb more CO2 until the optimum peak of 0.8 -1.0%. We will hopefully not see climate change-induced increases in carbon to this extent. For every doubling of CO2, C3 plants will improve their photosynthetic efficiency by 25-75 percent. However, soil nitrogen content must be increased to achieve higher biomass.
The temperature rises will increase photosynthesis. However, plants will transpire more and close their stomata to reduce transpiration. Plants can grow deeper roots to access more water and maintain the potential gradient. Or roll their leaves to reduce surface area and develop thicker leaf wax to reduce solar interception.
4. How do different environmental stresses, such as drought or high salinity, affect photosynthesis in plants, and how can we mitigate these effects?
Drought will directly reduce photosynthesis by reducing stomatal conductance to reduce transpiration. Other indirect effects can be plant growth inhibition or shedding leaves to minimize water use and the photosynthetic area. Increasing irrigation can help crop plants cope with physiological changes and improve photosynthetic rate. Scientists are also developing drought-tolerant or resistant cultivars by focusing on morphological or physiological parameters to improve water use efficiency.
Salinity affects photosynthesis by reducing chlorophyll content, leaf area, stomatal conductance, and photosystem II efficiency. Using salt-tolerant plants, improving farm management, partial root-zone drying, drip, micro-jet irrigation, and leaching salts from the root zone will prevent salinity.
Salinity is increasing due to low precipitation, higher evaporation through higher temperatures, irrigation through saline water, and poor cultivation practices. Several common factors cause drought and salinity; addressing them can prevent adverse effects on photosynthesis. Drought and salinity can reduce crop yields by 20-50 percent.
5. How do different types of plants (e.g., C3, C4, CAM) differ in their photosynthetic pathways?
Plant anatomical, physiological, and efficiency variations accompany the difference in photosynthetic pathways C3, C4, and CAM.
Most plants and crops are C3 plants, such as rice, wheat, potato, cowpea, cassava, and soybean. They open their stomata to let in CO2 but also let out water vapor, so water use efficiency is reduced. Therefore C3 plants are disadvantaged in the warmer climate, where C4 plants are more abundant. Due to more photorespiration photosynthetic efficiency of C3 plants is lower. Photosynthetic efficiency in C3 plants is only 4.6 percent, so scientists want to improve the efficiency to improve the productivity of crop plants. However, the leaves are anatomically simpler, and the photosynthesis process has fewer steps, so C3 plants require less energy and are the abundant plant type.
C4 plants are an adaptation to thrive in warmers regions because plants tend to close their stomata to cut water vapor loss. However, this reduces the CO2 available for fixation. So C4 plants ensure adequate photosynthesis occurs by avoiding photorespiration. Consequently, C4 plants’ photosynthetic efficiency is 50 percent higher than C3 plants and is around 6 percent. Examples of C4 plants are maize, sorghum, and sugarcane.
CAM plants usually have no true leaves, which have been reduced to spines. These include succulents such as opuntia and sedum, epiphytes like orchids, and bromeliads like pineapple. CAM plants are metabolic flexible; seedlings and well-watered adults switch to C3 photosynthesis. In comparison, water and salinity stress conditions can induce CAM.
6. How do different pigments, such as chlorophyll and carotenoids, play a role in photosynthesis?
Chlorophyll, the green pigment, is crucial for photosynthesis, as it is the phytochemical that absorbs solar light. It is also the most abundant pigment in plants. There are two types of chlorophyll molecules: chlorophyll-a and chlorophyll-b.
- Chlorophyll-a is the one necessary for photosynthesis. It accounts for ¾ of the chlorophyll in a plant. It absorbs 429 nm (violet-blue) and 659 nm (orange-red) wavelengths; see Figure 3.
- Chlorophyll-b is not present in all plants, and when it does occur, it makes up about ¼ of the total chlorophyll. It absorbs light at 455 nm (violet-blue) and some at 642 nm (orange) and passes it on to Chlorophyll-a.
Without chlorophyll, there would be no photosynthesis. The pigment is present in chloroplasts and is needed for both the stages of photosynthesis – carbon fixation and the Calvin-Benson Cycle.
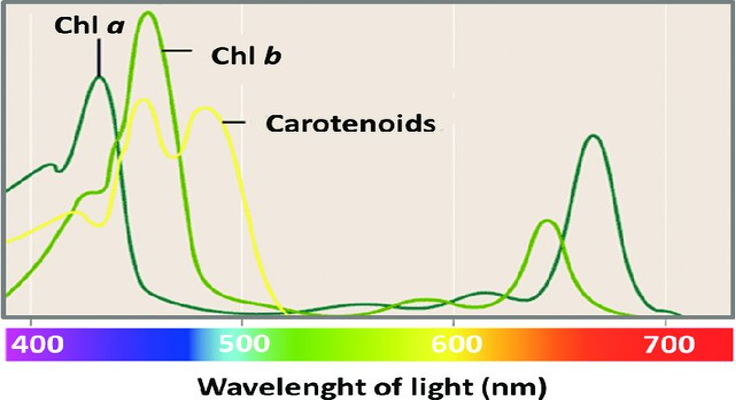
Figure 3: “Chlorophyll a, b and carotenoids absorbance spectra,” Guidi et al. 2017. (Image credits: doi: 10.5772/67887)
Carotenoids are yellow, orange, red, and purple pigments essential for photosynthesis. Carotenoids absorb a wide range of wavelengths, from 400–550 nm (violet to green), and pass the absorbed energy to chlorophyll. Hence they improve photosynthetic efficiency. Another essential function is to protect against photoinhibition due to exposure to excessive light. The triplet-triplet energy transfer from chlorophylls to carotenoids helps in photoprotection. In bacteria, carotenoids also provide structural stabilization for the photosynthetic apparatus.
7. How has our understanding of photosynthesis evolved, and what are some current active research areas in this field?
It was in the 1700s that photosynthesis was first discovered.
- Jean Senebier observed that plants fixed air in the presence of sunlight.
- Joseph Priestley showed that plants made air breathable and is credited with discovering oxygen.
- Jam Ingenhousz demonstrated that this phenomenon occurred in sunlight and green plant parts.
- Lazzaro Spallanzani showed that plants form “good air” (oxygen) from fixed air (carbon dioxide).
In the 1800s, Theodore De Saussure showed that plants absorb carbon dioxide and that oxygen is produced as an end product. He was the first to propose that carbon was taken from the air. Saussure explained the source of each significant nutrient used by plants and concluded water drawn from the roots was the source of hydrogen and oxygen.
It was in 1915 that Hans Kniep made the first measurement of photosynthesis. He measured the photosynthesis rate through gas exchange in the bubble method. It is the method still used to demonstrate photosynthesis in school laboratories.
Currently, there are several exciting developments in photosynthesis research, including finding new kinds of photosynthesis like artificial photosynthesis, understanding reactions, improving C3 and C4 plants’ performance, improving light storage by plants, reducing photorespiration, increasing rubisco content, understanding effects of climate change on photosynthesis, improving photosynthesis in precision agriculture, etc.
8. What role do photosynthetic bacteria play in the ecosystem?
Cyanobacteria, or blue-green algae, which live anaerobically and aerobically, were the first photosynthetic organisms that have lived since 3.5 billion years ago. About 1.2 billion years later (2.3 billion years ago), their oxygen had accumulated enough to be a significant atmospheric gas, making aerobic life possible.
Terrestrial and higher plants carry out most photosynthesis, but their respiration demand on O2 leaves little for the atmosphere, around 0.1 percent. Therefore, most of the current atmospheric O2, about 99 percent, is produced by marine cyanobacteria and eukaryotic algae. Prokaryotic bacteria produce another 1 percent of oxygen. Cyanobacteria are also responsible for fixing nitrogen in the seas.
Photosynthetic bacteria are also vital primary producers in marine ecosystems and serve as food to other trophic levels, including small and big animals.
Photosynthetic bacteria are important soil nitrogen fixers used as biofertilizers to improve soil and agricultural production.
9. What are some potential applications of photosynthesis research (e.g., biofuels, sustainable agriculture)? How can we use photosynthesis as a model for developing new technologies that convert light energy into useful products?
Some of the potential applications of photosynthesis are:
- The use of algal photosynthesis to produce biofuels, chemicals, and biobased products for the energy crisis or new photosynthetic batteries for microprocessors for IoT (Internet of Things)
- Medicinal applications such as a potential cancer cure or using products of bacterial photosynthesis, such as coenzyme Q10, for treating heart disease, anemia, and
brain vascular injury etc.
- Artificial photosynthesis has several applications, such as making photovoltaic panels for solar energy
- New systems like the hybrid biological-inorganic photosynthesis system for more food production
- Photocatalysis to treat plastic wastes
- Use of nitrogen fixers and other beneficial microbes as biostimulants, biofertilizers, and biocontrol in sustainable agriculture
10. How can we use advances in synthetic biology to engineer photosynthetic organisms for improved efficiency or novel applications?
Scientists use photosynthetic microbes as synthetic biology hosts for utilizing solar energy to optimize food, fuel, and fiber production. The most common bacteria used are cyanobacteria, diatoms, chlorophytes, and eustigmatophytes. The number of studies on the subject has increased in the past few years. Though there are not enough genetic engineering tools for microalgae compared to heterotrophic hosts, CRISPR could be a helpful precision editing and advanced genetic engineering tool.
Measuring Photosynthesis
After scientists have chosen how they will conduct their experiments on photosynthesis and the themes they want to cover, they must select the devices that will help them in the analysis. Non-destructive, accurate, rapid results are crucial, just as it is necessary to be able to analyze photosynthesis in the field. Portable tools like the C-340 Handheld Photosynthesis System that can measure photosynthesis and other gas exchanges like transpiration, stomatal conductance, and respiration can be a great asset. Moreover, the tool can also measure chlorophyll fluorescence and covers many of the modern methods of photosynthesis analysis for plant science research.
Sources
C3 and C4 photosynthesis. Student Materials. (2018, January 11). Retrieved March from https://serc.carleton.edu/integrate/teaching_materials/food_supply/student_materials/1167
Cui, H. (2021). Challenges and Approaches to Crop Improvement Through C3-to-C4 Engineering. Frontiers in Plant Science, 12. https://doi.org/10.3389/fpls.2021.715391
Demmig-Adams, B., Cohu, C. M. & Adams III, W. W. (2012) Dealing With the Hazards of Harnessing Sunlight. Nature Education Knowledge 4(1):18
Guidi, L., Tattini, M., & Landi, M. (2017). How Does Chloroplast Protect Chlorophyll Against Excessive Light? InTech. doi: 10.5772/67887
Hashimoto, H., Uragami, C., & Cogdell, R. J. (2016). Carotenoids and Photosynthesis. Sub-cellular biochemistry, 79, 111–139. https://doi.org/10.1007/978-3-319-39126-7_4
Kasting, J. F.; Siefert, J. L.; Life and the Evolution of Earth’s Atmosphere, Science, 2002, 296, 1066.
Kropp, H., & Halasey, A. (2014, August 02). CAM Plants. ASU – Ask A Biologist. Retrieved from https://askabiologist.asu.edu/cam-plants
Leegood, R. (2013). Photosynthesis. Encyclopedia of Biological Chemistry, 492-496. https://doi.org/10.1016/B978-0-12-378630-2.00049-9
Light-dependent reactions. Light-Dependent Reactions – an overview | ScienceDirect Topics. (n.d.). Retrieved from https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/light-dependent-reactions#
Meacham-Hensold, K. (2020, March 18). The difference between C3 and C4 plants. Retrieved from https://ripe.illinois.edu/blog/difference-between-c3-and-c4-plants
Shrivastava, P., & Kumar, R. (2015). Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi journal of biological sciences, 22(2), 123–131. https://doi.org/10.1016/j.sjbs.2014.12.001
Vavitsas, K., Kugler, A., Satta, A., Hatzinikolaou, D. G., Lindblad, P., Fewer, D. P., Lindberg, P., Toivari, M., & Stensjö, K. (2021). Doing synthetic biology with photosynthetic microorganisms. Physiologia plantarum, 173(2), 624–638. https://doi.org/10.1111/ppl.13455
Wang, C., Guo, L., Li, Y. et al. (2012)Systematic Comparison of C3 and C4 Plants Based on Metabolic Network Analysis. BMC Syst Biol 6 (Suppl 2). https://doi.org/10.1186/1752-0509-6-S2-S9
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science