November 16, 2021 at 4:27 am | Updated March 16, 2022 at 5:29 pm | 8 min read
Definition of Root Respiration
Plant respiration happens not only in the leaves, but also through the root system. In root systems, oxygen (O2) in soil pores diffuses into the root hairs and gets transported to all parts of the roots. Plant roots use photosynthates during respiration for their various functions, like nutrient uptake and maintaining and growing their tissues. The carbon dioxide (CO2) formed is then released into the soil, diffusing through the pores into the atmosphere; see Figure 1, terrestrial soils.

Importance of Root Respiration
Root respiration (Rr) has three components, maintenance, growth, and rhizospheric respiration.
- Maintenance respiration is the energy—generated by respiration—necessary to keep the roots alive.
- Root growth respiration is the part used to provide energy for growing roots and new tissues.
- Rhizospheric respiration is the respiration of microbes living in the rhizosphere—the soil zone around roots—where roots influence biological and chemical activities. The microbes in the rhizosphere, which use root exudates for their respiration, are involved in nutrient cycling and maintaining soil fertility.
Though root respiration helps in plant growth and productivity, it is also resource-intensive, and up to 75% of the assimilated carbon can be used up in many species. As a result, root respiration rates can affect above-ground growth.
Subscribe to receive our monthly round-up of articles.
Species Dependant Requirement

The oxygen requirements of plants will vary across species, where some need more than others. Most species cannot function in oxygen deficit soils.
Waterlogged soils are a common cause of oxygen deficiency. When water clogs soil pores, it hinders oxygen diffusion, causing an oxygen deficit. The result is that root water permeability is reduced. The entire plant will suffer from water deficiency, showing symptoms like leaf yellowing and wilt. Agronomists are familiar with this problem.
Most plants can survive a brief spell of waterlogging. When soils are waterlogged for a long duration, the roots and soil microbes use all the available oxygen. Under prolonged submergence in water, most plants’ roots are attacked by anaerobic pathogens, which replace aerobic bacteria in the soil or rot. The tissues get decomposed by anaerobic microbes and produce methane and carbon dioxide; see Figure 1, flooded soils.
Since plants respire less during the cold winter months, waterlogging causes less harm in this season. However, because roots respire more during summer, waterlogging quickly leads to oxygen deficit.
Some species whose roots grow in soggy conditions have evolved different adaptations to get oxygen.
- Wetland plants have aerenchyma and an impermeable barrier to prevent oxygen loss through the roots into the soil. The plants send oxygen through intracellular aerenchyma pathways to the sources. For example, this is how rice (Oryza sativa) roots respire in waterlogged conditions.
- Some mangrove trees grow pneumatophores or breathing roots, which have lenticels through which they absorb oxygen when submerged under seawater during high tides, see Figure 2.
Anaerobic Respiration
At times even species adapted to waterlogging can suffer in oxygen-deficient conditions, resulting in anoxia in roots, especially if the whole plant is submerged during floods. In these cases, anaerobic respiration begins within roots, and the plant goes into survival mode, carrying on only the processes necessary to live. If anoxia continues, anaerobic respiration will lead to plant decay.
Global Importance of Root respiration
Root respiration’s importance cannot be overstated. The process contributes to an average of 50% of soil respiration, an essential component of the global carbon cycle.
The actual, case-by-case contribution can vary between 10 and 90% of the soil respiration, based on differences in ecosystem and climate. For example, annual root respiration in cool temperate forests is 4.8 t C ha−1, around 45% of the total yearly soil respiration (10.6 t C ha−1 year−1).
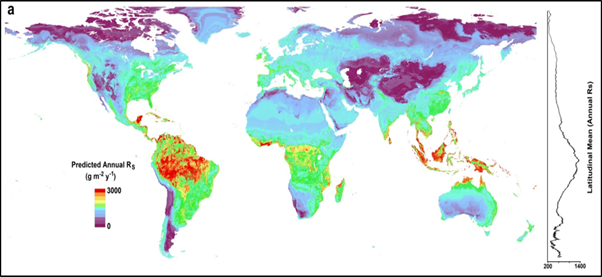
Soil respiration transfers carbon from the soil to the atmosphere and is the second-largest carbon flux (50-75 Pg C/yr), following gross primary production (100-120 Pg C/yr). Carbon flux through soil respiration is higher than the annual global terrestrial net primary productivity (NPP) of 50-60 Pg C/yr. See Figure 3 for global variations of soil respiration.
Hence, even a tiny change in root respiration can be consequential for the global carbon cycle. Both rising levels of CO2 and temperature can increase root respiration, which already exceeds NPP. So any gain in carbon sequestration through rising CO2 can be reversed by even small increases in root respiration.
Factors Affecting Root Respiration
Root respiration rates are crucial because respiration involves the loss of carbohydrates, which plants use for growth and biomass accumulation. While root respiration is vital, very high rates can impact plant productivity. Therefore, various intrinsic and environmental factors can impact plants by affecting root respiration.
Some of the critical environmental factors are:
Soil structure: A good soil texture that holds enough water but has proper drainage is necessary for root respiration. Heavy clay or compacted soil that stores water and has less drainage will impact root respiration by reducing soil oxygen availability.
Soil temperature: An increase in soil temperature raises total root respiration and specific root respiration (rate per unit of mass of roots). Species and cultivars that can tolerate prolonged heat without raising their root respiration can instead use their resources for root elongation, nutrient uptake, and membrane stability. Also, as soil temperature increases, the oxygen concentration in soil water decreases.
Soil nitrogen content: Increasing nitrogen in the soil lowers root respiration, but the form nitrogen takes in the soil can be critical. Overall, there is less bacteria when nitrates are present, so rhizosphere respiration—including bacterial respiration—falls. However, nitrogen as ammonium attracts more bacteria and increases rhizosphere respiration.
Increasing soil nitrogen can enhance fine root growth, which results in higher specific root respiration. As a result, carbon allocation to roots is lower, but fine root turnover can rise and increase carbon flux to the soil; however, the impact of nitrogen on fine root turnover is not completely clear.
Soil Potassium: The presence of soil potassium is crucial for root respiration rate and its effects on the roots. Absence of potassium can lead to increased root respiration and soils high in potassium, but low in oxygen can hinder roots’ ability to absorb water. Low potassium, however, has not been seen to affect water permeability.
Atmospheric CO2: As CO2 levels rise in the atmosphere, the CO2 in the soil also increases. Scientists accept that new root growth is increased with higher levels of soil CO2, which can be seen in higher total root respiration.
Seasons: Temperature is the main reason behind the seasonal variation in root respiration, with higher rates in summer and lower rates in winter. However, the extent of seasonal influence can vary depending on the ecosystem.
Intrinsic factors in the root, which can influence root respiration rates are:
Root nitrogen: All studies agree that respiration rate increases as nitrogen content in root tissue increases.
Root density: Across 245 plant species, both woody and non-woody, respiration rates decreased as the density of the root tissue increased.
Root types: Historically, scientists thought that all parts of the roots did not respire at the same rate. Coarse and medium roots had the same respiration rates, but thinner roots with higher nitrogen content would respire faster. However, a recent metastudy of 52 studies covering 254 woody and non-woody species, found that specific root length and root diameter did not influence root respiration rates. The study did not include many tropical ecosystems, so the conclusion is not global.
Growth form: Broad-leaved trees have higher respiration rates than conifers. The scientists reasoned that broadleaved species have higher leaf area and, therefore, photosynthesis and more carbon. This higher carbon supports increased respiration. There is no difference between root respiration between wood and non-woody species in their respiring rates.
Applications of Root Respiration
The study of root respiration has many applications:
- Breeding crops that can tolerate waterlogging
- Recommending irrigation parties for field crops and greenhouse cultivation
- Studying the effect of climate change on crops and their productivity
- Tracking the impact of nitrogen fertilizers on crop productivity
- Greenhouse management
- Research into soil carbon formation
- Soil carbon flux calculation
- Global carbon budget estimation
- Forest management through fertilization
- Soil management for crops and forests
Measuring Root Respiration Effects
Many scientists have noted that different estimation methods give varying results. The Infra-red Gas Analyzer in combination with closed chamber systems is popular but not the only method used. Measuring factors like leaf area or impact on root growth are more standard and non-destructive, using tools from CID Bio-Science Inc. It is easier to measure biomass accumulation and predict the effect of the environment on crops. However, we need to measure the process accurately to estimate its global impact due to climate change.
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Sources
Ben-Noah, I., & Friedman, S. P. (2018). Review and evaluation of root respiration and of natural and agricultural processes of soil aeration. Vadose Zone Journal, 17(1), 170119. https://doi.org/10.2136/vzj2017.06.0119
Blume H-P, Hartge KH, Schwertmann U (2007) Die Bedeutung des Ferdinand Enke
Verlages für die Verbreitung bodenkundlichen Wissens. In: Blume H-P, Stahr K (Hg)
Zur Geschichte der Bodenkunde (Chapter 4). Hohenheimer Bodenkundliche BerichteNr. 83
CNRS. (2016, Sept 15). How plant roots sense and react to soil flooding. Retrieved from https://www.cnrs.fr/en/how-plant-roots-sense-and-react-soil-flooding
Colmer T.D., Greenway H. (2005) Oxygen Transport, Respiration, and Anaerobic Carbohydrate Catabolism in Roots in Flooded Soils. In: Lambers H., Ribas-Carbo M. (eds) Plant Respiration. Advances in Photosynthesis and Respiration, vol 18. Springer, Dordrecht. https://doi.org/10.1007/1-4020-3589-6_8
Jia, S., Wang, Z., Li, X. et al. (2010). N fertilization affects on soil respiration, microbial biomass and root respiration in Larix gmelinii and Fraxinus mandshurica plantations in China. Plant Soil 333, 325–336. https://doi.org/10.1007/s11104-010-0348-8
Han, M., & Zhu, B. (2020). Linking root respiration to chemistry and morphology across species. Global Change Biology, 27(1), 190–201. https://doi.org/10.1111/gcb.15391
Lak, Z.A., Sandén, H., Mayer, M. et al. (2020). Specific root respiration of three plant species as influenced by storage time and conditions. Plant Soil 453, 615–626. https://doi.org/10.1007/s11104-020-04619-9
Larionova, A.A., Sapronov, D.V., Lopez de Gerenyu, V.O. et al. (2006). Contribution of plant root respiration to the CO2 emission from soil. Eurasian Soil Sc. 39, 1127–1135. https://doi.org/10.1134/S1064229306100103
Lee, J. S. (2018). Relationship of root biomass and soil respiration in a stand of deciduous broadleaved trees—a case study in a maple tree. J Ecology Environ 42, 19. https://doi.org/10.1186/s41610-018-0078-z
Lee, M.-sun, Nakane, K., Nakatsubo, T., & Koizumi, H. (2005). The importance of root respiration in annual soil carbon fluxes in a cool-temperate deciduous forest. Agricultural and Forest Meteorology, 134(1-4), 95–101. https://doi.org/10.1016/j.agrformet.2005.08.011
Nakamura, M., Noguchi, K. Tolerant mechanisms to O2 deficiency under submergence conditions in plants. J Plant Res 133, 343–371 (2020). https://doi.org/10.1007/s10265-020-01176-1
Pregitzer, K. S., Zak, D. R., Loya, W. M., Karberg, N. J., King, J. S., & Burton, A. J. (2007). The contribution of root – rhizosphere interactions to biogeochemical cycles in a Changing World. Cardon, Z.G. & J. L. Whitbeck. The Rhizosphere, 155–178. Academic Press, https://doi.org/10.1016/b978-012088775-0/50009-4
Raich, J. W. &. Schlesinger, W. H. (1992) The global carbon dioxide flux in soil respiration and its relationship to vegetation and climate, Tellus B: Chemical and Physical Meteorology, 44:2, 81-99, DOI: 10.3402/tellusb.v44i2.15428 To link to this article: https://doi.org/10.3402/tellusb.v44i2.15428
Schlesinger, W.H. 1977. Carbon balance in terrestrial detritus. Ann. Rev. Ecol. Cyst. 8, 51-81
Shibistova, O., Lloyd. J, Evgrafova, S., Savushkina, N., Zrazhevskaya, G., Arneth, A., Knohl, A., & Kolle, O. (2002) Seasonal and spatial variability in soil CO2 efflux rates for a central Siberian Pinus sylvestris forest. Tellus 54B, 552-567.
Trolldenier, G., & Von Rheinbaben, W. (1981). Root respiration and bacterial population of roots I. Effect of nitrogen source, potassium nutrition and aeration of roots. Zeitschrift Für Pflanzenernährung Und Bodenkunde, 144(4), 366–377. https://doi.org/10.1002/jpln.19811440405
Wang W, Guo J and Oikawa T. (2007). Contribution of root to soil respiration and carbon balance in disturbed and undisturbed grassland communities,
northeast China. J. Biosci. 32, 375–384
Warner, D.L., B.P. Bond-Lamberty, J. Jian, E. Stell, & R. Vargas. (2019). Global Gridded 1-km Annual Soil Respiration and Uncertainty Derived from SRDB V3. ORNL DAAC, Oak Ridge, Tennessee, USA. https://doi.org/10.3334/ORNLDAAC/1736
Waterlogging and flooding. Retrieved from https://www.rhs.org.uk/advice/profile?PID=235
Xu, M., & Shang, H. (2016). Contribution of soil respiration to the global carbon equation. Journal of Plant Physiology, 203, 16–28. https://doi.org/10.1016/j.jplph.2016.08.007
Rachmilevitch, S., Lambers, H., Huang, B. (2006). Root respiratory characteristics associated with plant adaptation to high soil temperature for geothermal and turf-type Agrostis species, Journal of Experimental Botany, 57(3), 623–631. https://doi.org/10.1093/jxb/erj047
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






