October 14, 2020 at 3:37 am | Updated October 14, 2020 at 3:37 am | 7 min read
According to a review by Dr. Crocombe, hand-held spectrometers have historically been used by only small groups of people, such as analytical and drug companies. These days, spectroscopy is available in simple low cost devices for a multitude of applications for scientists and non-scientists alike. One of the most prominent uses of spectroscopy has been the use of the technology on leaves for plant research and agriculture.
Spectrometers – The Quick Background
Every element and compound has a distinct spectral signature. Elements and compounds will absorb, transmit (or allow light through), and reflect different light wavelengths in particular patterns based on their composition.
Light is made of different wavelengths measured in nanometers (or nm). So, spectrometers use white light and split it to produce a desired spectrum of light, which is then directed at an object. The spectrum dispersed by the object is then analysed to see what part was absorbed, transmitted, and reflected.
Subscribe to receive our monthly round-up of articles.
Spectrometers usually use a specific portion of the light, such as visible (400-700 nm), shortwave near infrared (~700-1050nm), infrared (~1000-1700nm), near infrared (~1200-2500nm), ultraviolet (<250nm), etc.
Portable spectrometers are miniature avatars of larger pieces of equipment, and provide data and help in analysis of plants in the field, laboratory, or classroom.
Portable spectrometers are designed to be rugged, easy to use, and require no extensive expertise. They can withstand a wide range of environmental conditions, and many can be connected directly to wifi/computer to record and analyze data. Also, the readings are precise and rapid.
Applications of Spectrometers
Though all compounds show spectroscopy, pigments have the strongest spectral signatures, as they absorb most of the light due to their color.
So, leaf spectrometers use a wide range of light—from visible to near infrared—in plant research and agriculture to measure chlorophyll, anthocyanin, and carotenoids, as well as the water content in leaves.
The importance of these compounds to the plants and the reasons for measuring them are explained below –
The different colors of the three pigments chlorophyll, anthocyanin, and carotenoids. (Image credits: https://byjus.com/biology/pigments/)
Chlorophyll
Nitrogen, one of the major nutrients needed by plants, is needed to produce chlorophyll. Nitrogen levels are directly correlated with amounts of chlorophyll. The quantity of chlorophyll—the green pigment in leaves, in turn, determines the amount of photosynthesis that takes place. Photosynthesis is a process in which sunlight captured by chlorophyll is used to combine carbon dioxide from the air with water absorbed by the roots to make simple sugars, the basic food for plants.
Nitrogen is important for plants; its deficiency causes leaves to lose their green color and results in reduced protein and starch. As a result, growth, health, flowering, and fruiting in plants are all reduced. Thus, both quality and quantity of a crop can be affected by nitrogen levels.
Anthocyanins
The red and orange colors in leaves, flowers, and fruits are due to the presence of anthocyanins. They are usually present in leaves, but higher concentrations of chlorophyll usually mask them.
Anthocyanins are important for the defense of plants. In the leaves, they help plants in camouflage and to repel herbivores and parasites. The pigment also helps the plant tolerate stress, such as drought, UV radiation, and heavy metals. Moreover, anthocyanins protect chloroplasts that contain chlorophyll from damage by strong sunlight. The red pigment also scavenges free radicals and reactive oxygen species and protects the plants. Overall, it is vital for the survival of the plant.
Carotenoids
This pigment colors leaves, flowers, and fruits yellow. In the leaves, carotenoids help plants absorb the light energy needed for photosynthesis to occur. Like anthocyanins, they too are antioxidants and reduce free radicals that could damage the leaves. Carotenoids are also an indicator of water stress in plants.
Water Content
Plants and trees have seventy-seven to ninety-one percent of water content, making it the highest component in the tissues. It is one of the ingredients used in photosynthesis to manufacture food. Water is also how most of the nutrients from the soil and the food manufactured in the leaves are moved around in the plants. It is necessary for germination and growth, besides regulating the temperature in plants.
Water also has a strong spectral signature and is therefore useful in studying plant health, water stress, and soil water availability.
Leaf Spectrometers in Agriculture
In agriculture, horticulture, viticulture, and silviculture, leaf spectrometers are used to improve crop growing conditions and monitor plant health. The following examples of leaf spectrometer uses in agriculture show how it can be beneficial to farmers.
Nitrogen as an Indicator of Plant Growth and Yield
Leaf spectrometers are used to detect and quantify the amounts of chlorophyll in leaves with the help of several formula. Repeated measurements at different stages provide real time information on several aspects of plants, such as the following:
- The amount of nitrogen in plants, which is an indicator of plant health.
- The level of proteins in the leaves, which is a reflection of proteins in grains or fruits. For example, this is important for wheat, whose prices depend on protein percentage.
- The efficiency of fertilizer use by plants, which shows the amount of nitrogen available to plants, which in turn, determines the concentrations of chlorophyll and proteins. Measurements at different stages can help the farmer fine-tune the quantity and frequency of fertilizer applications. This is a critical feature of precision agriculture.
Carotenoids to Detect Water Stress
Since levels of carotenoids decrease during times of water stress, the quantification of these pigments can help farmers in the early detection of drought. Reduced carotenoid levels affect plant development, resistance to stress, and photosynthetic activity. Researchers have established the relation between carotenoids and water stress in African eggplants (Solanum aethiopicum and S. macrocarpon), two important commercial crops. The resultant conditions and symptoms of plants, as shown in Figure 2, can be shared with farmers, as indicators of drought, to help them in farm management.

Figure 2: “The picture of African eggplant (RV100332) accession (A) Water stressed (B) Control treatments”, Mibei et al.2016. (Image credits: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5217938/)
Carotenoids to Monitor Effect of Growth Regulators
Foliar growth regulators can influence the levels of carotenoids in the leaves of cash crops, such as cashew (Anacardium occidentale L.) and can be used to monitor the efficacy of foliar applications of these supplements.
Leaf Spectrometers in Plant Research
All applications of leaf spectrometers in agriculture are initially tested by research before they can be used on farms. Also, leaf spectrometers have other uses in plant research, as shown by the examples below.
Measuring Foliar Water Content
Scientists have shown that the water content in leaves rises and falls with water availability in the soil, using handheld spectrometers in two coniferous pines species (See Figure 3). This suggests that simple-to-use handheld spectrometers can be of use in forestry and can replace expensive soil tests and satellite data to monitor drought stress in forests.
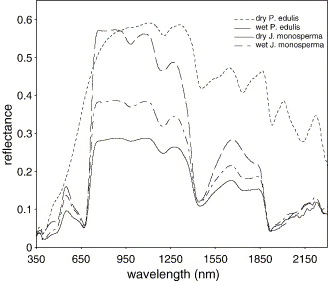
Figure 3: Spectral signatures of different levels of foliar water in two conifers Pinus edulis and Juniperus monosperma, Stimson et al. 2005. (Image credits: 10.1016/j.rse.2004.12.007)
Studying Plant Physiology
The relative content of chlorophyll, anthocyanins, and carotenoids, differ through plant development and age, such as leaf senescence and fruit formation. It is, therefore, necessary to track changes in the concentrations of the pigments over time. In a US study, non-destructive and inexpensive leaf spectrometers were used as the tools for monitoring pigment changes in maple by quantifying their reflectance. The scientists were able to determine the specific wavelengths to measure each pigment, a finding they hope can also be applied in remote sensing.
Developing Heat-Resistant Varieties
Since wheat is susceptible to heat, scientists have been attempting to develop varieties that can better withstand higher temperatures. The thickness of wax on leaves is instrumental in protecting leaves from heat stress. Therefore, optimising the leaf cuticle is one way to reduce excessive irrigation. Conventional methods of cuticle thickness are tedious. Instead, scientists tried and succeeded in estimating thickness of wax coating on leaves by measuring light absorbance and transmission. They found that the visible spectrum of light provided the most accurate estimate of leaf wax thickness.
The CI-710 Leaf spectrometer
The CI-710 Miniature Leaf Spectrometer is one of the most popular spectroscopic devices in plant physiology for use in fields, classrooms, and the laboratory. The CI-710 uses the Avantes Mini2048CL (~380-1100 nm) that covers a wide spectrum of light-visible, shortwave near infrared, and a part of near infrared spectra.
The CI-710 is used to measure leaf spectroscopy in the field, laboratories, and classrooms. It measures reflectance, absorbance, and transmission of light by leaves. It has a clip to hold the leaves and accompanying software that can be loaded on the tablet/computer. The unit can be attached to the tablet by a cable.
To use the spectral results, there are 29 pre-programmed indices and users can also add their custom indices.
Spectrometers Use in Agriculture and Research is Increasing
In 2016, small handheld spectrometers worth $170 million were sold, and by 2021, the sales are expected to rise to $297 million. The niche that this technology has carved out for itself is new and supplements the use of traditional large equipment in institutions, suggesting that spectroscopy constinues to develop new consumers and untapped applications.
—
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Featured blog image courtesy of Chanel Mason.
Source
Bojović, B., & Marković, A. (2009). Correlation between nitrogen and chlorophyll content in wheat (Triticum aestivum L.). Kragujevac J. Sci. 31:69-74. Retrieved from http://www.pmf.kg.ac.rs/kjs/volumes/kjs31/kjs31bojovicmarkovic69.pdf
Boyer, S. (2016, Jan 1). How much water is there in a tree? Is there any study that has measured water content in whole trees? Retrieved from https://www.researchgate.net/post/How_much_water_is_there_in_a_tree_Is_there_any_study_that_has_measured_water_content_in_whole_trees
Crocombe, R.A. (2018). Portable Spectroscopy. Applied Spectroscopy.72: 1701-1751. DOI:https://doi.org/10.1177%2F0003702818809719.
Fatima Camarillo-Castillo, F., Tattaris, M., Hays, D.B., & Reynolds, M.P. (2017, March 23).
Prediction accuracy of high-resolution spectral information for nondestructive phenotyping of epicuticular wax in wheat. Proceedings of the 3rd International TRIGO Wheat Yield Potential. 7-13. Retrieved fromhttps://repository.cimmyt.org/xmlui/bitstream/handle/10883/18162/58466.pdf?sequence=1&isAllowed=y#page=17
Gould K. S. (2004). Nature’s Swiss Army Knife: The Diverse Protective Roles of Anthocyanins in Leaves. Journal of biomedicine & biotechnology, 2004(5), 314–320. doi:10.1155/S1110724304406147
Lienard, S. & Torrens, K. (2019, September 23). In the leaves, they help plants in camouflage and to repel herbivores and parasites. Retrieved from https://www.bbcgoodfood.com/howto/guide/what-are-anthocyanins-and-why-are-purple-foods-so-healthy
Mibei, E. K., Ambuko, J., Giovannoni, J. J., Onyango, A. N., & Owino, W. O. (2016). Carotenoid profiling of the leaves of selected African eggplant accessions subjected to drought stress. Food science & nutrition, 5(1), 113–122. doi:10.1002/fsn3.370
Whiting, D. (2017, April 24). What is a Spectrometer? Sciencing. Retrieved from https://sciencing.com/spectrometer-5372347.html
Simcha Lev-Yadun, S., & Gould, K.S. (2009). Role of Anthocyanins in Plant Defence. Anthocyanins. Springer Science+Business Media, LLC. DOI: 10.1007/978-0-387-77335-3_2
Stimson, H.C., Breshears, D.D., Ustin, S.L., & Kefauver, S.C. (2005). Spectral sensing of foliar water conditions in two co-occurring conifer species: Pinus edulis and Juniperus monosperma. Remote Sensing of Environment, 96, 108-118. DOI: 10.1016/j.rse.2004.12.007
TNAU Agritech Portal. Role of water for growth and development of crops. Retrieved from http://agritech.tnau.ac.in/agriculture/agri_irrigationmgt_roleofwater.html
Ramapura, Ravikumar, R.L., Adiga, J., Kalaivanan, D., & Halesh, G.. (2017). Effect of plant growth regulators on leaf area, chlorophyll content, carotenoids, stomatal count, and yield of cashew (Anacardium occidentale L.) var. Bhaskara. Journal of Plantation Crops. 45. 141. 10.19071/jpc.2017.v45.i2.3309.
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






