April 29, 2024 at 5:15 pm | Updated April 15, 2026 at 2:24 am | 9 min read
- Fine roots have a diameter of 0.1–2 mm and are essential for nutrient acquisition for individual plants and carbon and nutrient cycling for ecosystems.
- Research shows fine roots are not a single group; root order determines form, anatomy, physiology, and function.
- Fine root age is another significant factor that influences functioning and dynamics.
Though fine roots are essential for plants’ ecological processes, less is known about them and their drivers than aboveground vegetation. Minirhizotron studies of fine roots are a standard method scientists use to fill this lacuna. In this article, you will learn how the tool can help characterize fine root production, age, growth patterns, turnover, mortality, lifespan, and carbon flux.
What are Fine Roots?
Fine roots have a diameter of 0.1–2mm and are involved in the absorption of water and nutrients by plants. They regulate and are crucial for natural ecosystems’ nutrient, carbon, and water cycles.
Minirhizotrons, cameras, and root image analytics have revealed information on the complexity of fine root systems. The number, length, biomass, color, branching, and mycorrhizal colonization differ within a population, between populations of a species, and between species and ecosystems.
Subscribe to receive our monthly round-up of articles.
So, fine roots are no longer seen as a single group below 2mm with common features and functions. What we have learned so far about fine roots in crops and perennial forest trees through minirhizotron systems is discussed below.
Fine Root Morphology
Morphology is one of the most studied aspects of fine roots. The traits studied are root length, diameter, branching, specific root length, root tissue density, and specific root surface area. These traits can show phenotypic plasticity and vary between species, variety, regions, and ecosystems, see Figure 1.
Morphological traits can be connected to functions and provide vast information on root and plant dynamics, such as mortality, turnover, biomass, production, and growth patterns, to understand drought strategies.
Fine roots can make up to 60% of the total root length in crops, but this depends on plant type and species. Generally, pulses have fewer fine and more coarse roots than cereals like wheat.
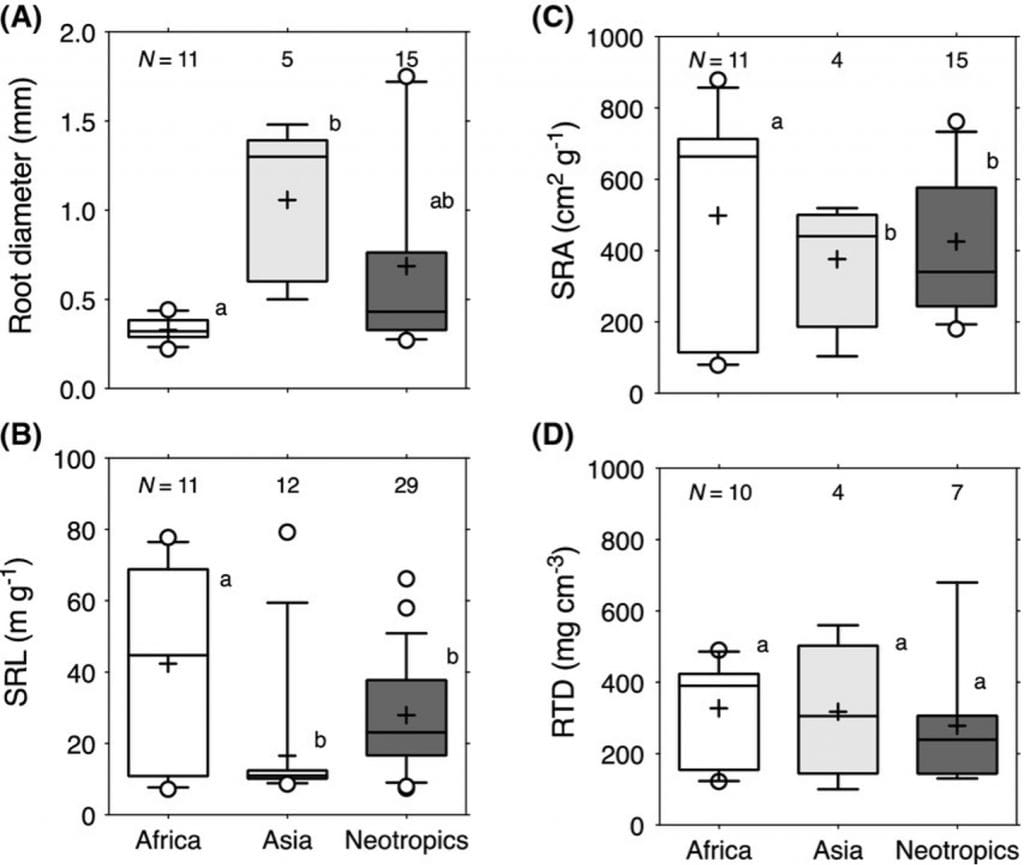
Figure 1: “Distribution of commonly measured fine-root morphological trait values in tropical forests in Africa, Asia and Neotropics (Central and South America). The total number of observations per region is indicated at the top of each box. RTD root tissue density, SRA specific root area, SRL specific root length,” Addo-Danso et al. 2020. (Image credits: https://doi.org/10.1007/s11258-019-00986-1)
Fine Root Order
While branching is a morphological trait, the position of an individual root in the branching hierarchy is called root order.
- Fine roots without dependent laterals are 1st order fine roots.
- Fine roots with one set of dependent laterals are 2nd order fine roots.
As the order increases, roots have more hierarchical levels of dependent laterals. Maximum acceptable root orders in woody perennials are three to four. First- and second-order fine roots comprise nearly 75% of fine roots. All fine roots begin as 1st order roots, and only a tiny subset grows laterals.
Fine roots of different order will vary in anatomy, morphology, physiology, function, and lifespan. See Figure 2. For example, as order increases, diameter and age increase.
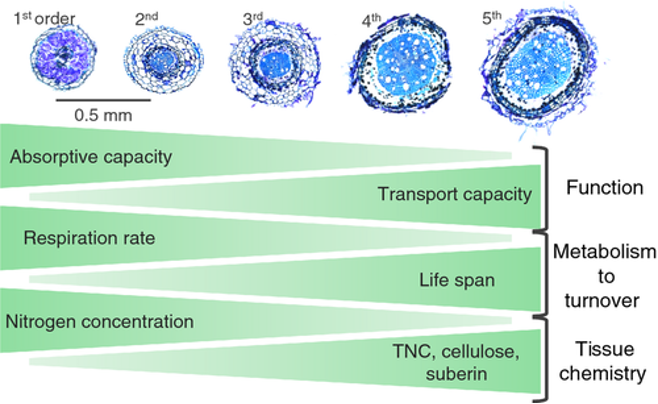
Figure 2: “Root cross-sections of Acer plantanoides show a typical pattern of increasing root diameter and secondary development with increasing root order. Notice that first- and second-order roots have little or no secondary development, and first- to third-order roots still possess intact root cortical cells, while fourth- and fifth-order roots have lost all cortex and instead have secondary xylem developed within the stele. Triangles depict simplified patterns of root function (absorptive and transport capacity) and root traits (respiration rate g−1 root, life span, total nonstructural carbohydrates (TNC) and other aspects of tissue chemistry) with increasing root order,” McCormack et al. 2001. ( Image credits: https://doi.org/10.1111/nph.13363)
Fine Root Production Patterns
The fine root production patterns differ among annual crops and perennial trees.
Earlier in the crop season, annuals have more young fine roots; towards the end, there are older ones. The production and growth patterns reflect more allocation to roots to enhance nutrient absorption in the early season and less later when more allocation is given to reproductive parts.
Fine roots production trends are more complex in woody perennials. Trees produce fine roots throughout the growing season with one or two peaks. Fine root peaks come after leaf and flower initiation. For example, apples and peaches have a first flush/peak of fine root production during or after blooming, followed by a second flush in early fall.
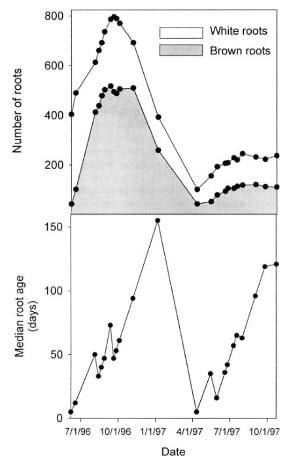
Figure 3: “Number of white and brown fine roots visible on 12 minirhizotrons in a West Virginia peach orchard from June 1996 through October 1997 (top panel) (data from Wells and others 2002a). Median root age of all fine roots visible on the minirhizotrons over the same interval (bottom panel),” Wells & Eissenstat. (2002). (Image credits: DOI: 10.1007/s00344-003-0011-1)
Fine Root Life Span
Minirhizotron images show that younger fine roots are white; as they age, they become brown (see Figure 3). Later, they turn black and shrivel before mortality. Minirhizotron studies also showed that fine roots did not increase in diameter or grow woody as they aged.
Annual crops that live less than a year have fine roots whose age differs slightly and lasts for a few months.
In contrast, forest perennials have a longer lifespan than annual crops or plants. The life period depends on the species. For example:
- Norway spruce have fine roots that live 250 to 350 days.
- Sixty-year-old sugar maples have fine roots that are 400 days old.
- Hardwoods and coniferous in Europe had fine roots with median lifespans over 930 days.
However, cultivated orchard woody trees have shorter lifespans. For example, fine roots life span in apples is 36-144 days, but in kiwifruits, it is only 28 days.
The longer lifespans of perennials and the yearly flushes produce complex and changing fine root age distribution. Moreover, the average age of fine roots changes at different times of the season.
Lifespans will also differ depending on root order. First-order roots have shorter median lifespans of 95 to 105 days, while second —and third-order roots have lifespans of 226-259 days.
Fine Root Function
Fine roots in perennials of similar diameter show different forms and functions. So, it is difficult to attribute a function based on single-pool diameter-based traits. Scientists agree that two factors affect functioning- age and root order- across species and sites.
There are two types- shorter-lived absorptive and longer-lived transport fine roots.
- Absorptive fine roots are lower-order roots that acquire and absorb nutrients and water.
- Transport fine roots belong to a higher root order. Their function is to transport the nutrients, providing structure and storage capacity.
The fine root traits associated with the two functions are detailed in Table 1.
In minirhizotron observations, installation can disturb existing transport fine roots. Therefore, minirhizotrons are effective only in monitoring absorptive fine-root dynamics that develop after root tube installation. On the other hand, isotopic techniques are biased towards higher-order transport roots with more mass.
Table 1: “Common traits associated with uptake and absorption of soil water and nutrients by fine roots or with increased capacity to efficiently transport resources within the root to more proximal plant tissues,” McCormack et al. 2001. (Image credits: https://doi.org/10.1111/nph.13363)

Function Change with Age
Even among absorptive fine roots, age matters.
- Nutrient Uptake: As fine roots age, their nutrient absorptive capacity reduces. The extent of decline depends on species and nutrients. For example, citrus fine roots have a lifespan (300 days) five times longer than apples (60 days). Phosphorus uptake by citrus is 400 pmol P g-1 s-1 against 2000 P g-1 s-1 by apples.
The decline mode can also differ; apples’ phosphorus uptake peaked in the first 25 days and gradually declined continuously. Meanwhile, citrus phosphorus uptake peaked in four days, was reduced by 35% in a week, and stabilized at that level for the entire lifespan.
- Respiration: Root respiration also declined with age in citrus and apples.
- Water uptake: Older roots have less hydraulic properties than younger roots. For example, younger white fine roots transport 4.2 to 97 mm3 cm-1 h-1 compared to 4.36 to 32 mm3 cm-1 h-1 in older brown
The nutrient and water uptake change can be due to anatomical and physiological processes. Scientists think more studies with minirhizotron observations will be necessary to understand the functioning of age- and order-based fine roots.
Fine Root Turnover
Though fine roots form a small part of underground living carbon, their turnover significantly contributes to soil organic carbon. Fine root production is around 22% of forest net primary production (NPP).
Turnover results from production, mortality, and decay of fine roots. Fine roots’ lifespan is shorter than other larger roots, so flux or addition of root carbon and nutrients is rapid. This is also why fine roots are crucial for ecosystem carbon and nutrient cycling.
Carbon Cycles
High fine root turnover is a significant contributor to carbon in soils. Therefore, it is necessary to accurately estimate root turnover and standing biomass.
Root respiration is another significant component of soil carbon flux, as roots account for 50% of soil respiration that adds carbon to the soil.
Studies on climate change’s effect on soil carbon indicate that as air CO2 increases, soil carbon increases. This area requires more research.
Advantages of Minirhizotrons Use
Soil cores, monoliths, sequential coring, minirhizotrons, in-growth cores, trenches, and bags/nets are the methods used to study fine roots. The estimation method affects parameters, so the choice of technique must be made carefully.
Minirhizotron systems are a nondestructive method of studying fine roots. For example, the root imagers, tubes, and software offered by CID Bioscience Inc. have long been a standard minirhizotron system scientists use. The company provides two imagers for nondestructive imaging in fields and forests- the CI-600 In-Situ Root Imager and the CI-602 Narrow Gauge Root Imager.
The Minirhizotron system can be used for estimating several fundamental fine root dynamics as discussed below:
- Minirhizotrons, which can be used for long-term studies, are suitable for establishing lifespans.
- Annual fine root turnover is difficult to estimate as production and death occur at the same time and place. It is best estimated by minirhizotron, albeit indirectly. It is calculated as the ratio of annual root length to average live root length or as the inverse of median root longevity.
- Fine root production and mortality can be measured using a combination of minirhizotrons and soil cores for roots of the same diameter class.
- Carbon and nutrient flux through acceptable root mortality is estimated indirectly by multiplying turnover with carbon and nutrients in fine root biomass.
Disturbance from root tube installations can last years and show more fine root productivity than mortality. So, some aspects of fine root dynamics can be overestimated in the initial study years.
Moreover, variations in the mode of use and data collected exist. For easier comparison of results, idiosyncratic units could be avoided. Using volumetric units will also make minirhizotron-based estimates comparable with aboveground measurements.
Minirhizotrons Are Essential for Fine Root Studies
Direct observation through minirhizotrons has revealed various below-ground fine root dynamics that have been a mystery for a long time. However, most studies so far have been short-term. Long-term studies will be necessary to monitor the lifespan and turnover of long-lived fine roots. Also, the mechanisms behind various observed trends are unclear. These gaps must be overcome to understand the overall functioning of the root system and plants.
Sources
Addo-Danso, S.D., Defrenne, C.E., McCormack, M.L. et al. (2020). Fine-root morphological trait variation in tropical forest ecosystems: an evidence synthesis. Plant Ecol 221, 1–13. https://doi.org/10.1007/s11258-019-00986-1
Espeleta, J. F., & Clark, D. A. (2007). Multi‐scale variation in fine‐root biomass in a tropical rain forest: A seven‐year study. Ecological monographs, 77(3), 377-404.
Finér, L., Ohashi, M., Noguchi, K., & Hirano, Y. (2011). Factors causing variation in fine root biomass in forest ecosystems. Forest Ecology and Management, 261(2), 265-277.
Johnson, M. G., Tingey, D. T., Phillips, D. L., & Storm, M. J. (2001). Advancing fine root research with minirhizotrons. Environmental and Experimental Botany, 45(3), 263-289.
McCormack, M. L., Dickie, I. A., Eissenstat, D. M., Fahey, T. J., Fernandez, C. W., Guo, D., … & Zadworny, M. (2015). Redefining fine roots improves understanding of below‐ground contributions to terrestrial biosphere processes. New Phytologist, 207(3), 505-518.
Nikolova, P. S., Bauerle, T. L., Haeberle, K. H., Blaschke, H., Brunner, I., & Matyssek, R. (2020). Fine-root traits reveal contrasting ecological strategies in European beech and Norway spruce during extreme drought. Frontiers in Plant Science, 11, 1211.
Neumann, M., Godbold, D. L., Hirano, Y., & Finér, L. (2020). Improving models of fine root carbon stocks and fluxes in European forests. Journal of Ecology, 108(2), 496-514.
Sun, L., Ataka, M., Han, M., Han, Y., Gan, D., Xu, T., … & Zhu, B. (2021). Root exudation as a major competitive fine‐root functional trait of 18 coexisting species in a subtropical forest. New Phytologist, 229(1), 259-271.
Wang, N., Wang, C., & Quan, X. (2020). Variations in fine root dynamics and turnover rates in five forest types in northeastern China. Journal of Forestry Research, 31(3), 871-884.
Wells, C. E., & Eissenstat, D. M. (2002). Beyond the roots of young seedlings: the influence of age and order on fine root physiology. Journal of Plant Growth Regulation, 21, 324-334. DOI: 10.1007/s00344-003-0011-1
Xing, Y., Chen, M., Dao, J., Lin, L., Chen, C., Chen, Y., & Wang, Z. (2024). Fine-root morphology of woody and herbaceous plants responds differently to altered precipitation: A meta-analysis. Forest Ecology and Management, 552, Article 121570. https://doi.org/10.1016/j.foreco.2023.121570
Yuan, Z. Y., & Chen, H. Y. (2010). Fine root biomass, production, turnover rates, and nutrient contents in boreal forest ecosystems in relation to species, climate, fertility, and stand age: literature review and meta-analyses. Critical Reviews in Plant Sciences, 29(4), 204-221.
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






