April 24, 2023 at 3:17 pm | Updated April 24, 2023 at 3:17 pm | 9 min read
The global carbon cycle is crucial as it supports the food chain of natural ecosystems and humans. People also depend on the carbon cycle for industrial systems. Excess greenhouse gases of anthropogenic origin, carbon dioxide, and methane, are now driving climate change. Like stomatal conductance, green plants’ role and processes are central to the carbon cycle. This article shows the various ways in which stomatal conductance is influencing the global carbon cycle and us.
The Carbon Cycle
The global carbon cycle involves exchanges of carbon between and within the four sinks, the atmosphere, land, oceans, and fossil fuels. Photosynthesis transfers carbon to plants from the atmosphere and is held in terrestrial ecosystems. Some photosynthates are transported to the roots and rhizosphere and become part of soil carbon. Photosynthesis by marine algae and bacteria provides food for all marine creatures.
Over millions of years, organic matter deposits in the soil and ocean have been fossilized. The fossil fuels we burn are the result of the carbon cycle in the past.
Subscribe to receive our monthly round-up of articles.
Anthropogenic activities like burning wood or fossil fuels will circulate carbon back into the atmosphere. Terrestrial plant respiration releases carbon dioxide back into the atmosphere. While decomposition processes both in soil and water will also give back carbon to the air, see Figure 1.
Stomatal conductance is essential in terrestrial systems as it regulates gas exchanges- photosynthesis and plant respiration.
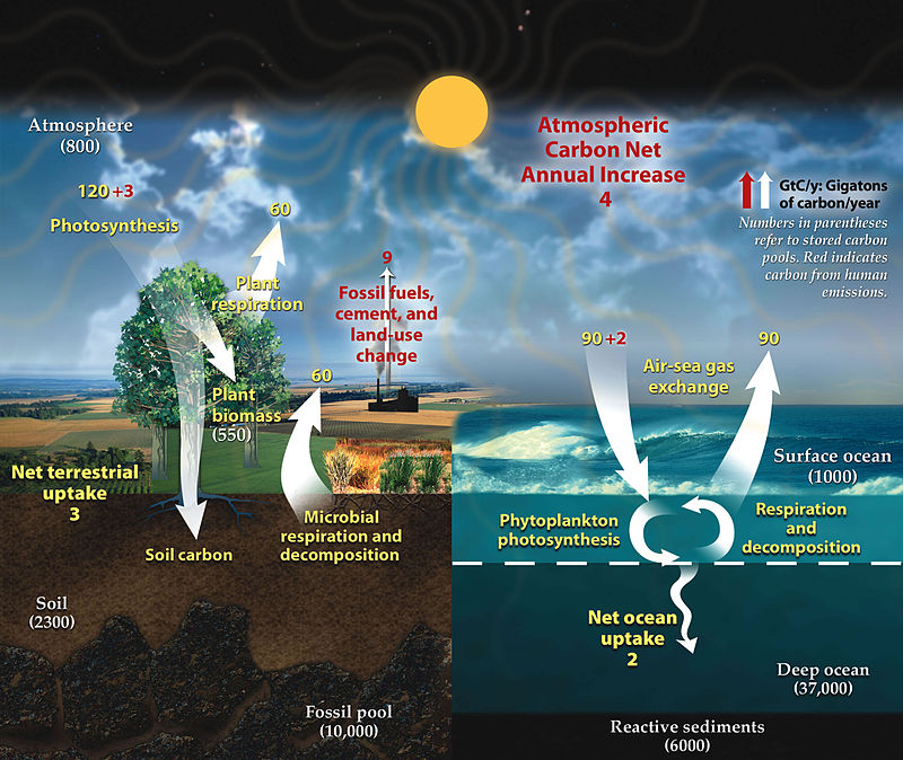
Figure 1: “The carbon cycle of the Earth. Numbers represent the mass of carbon in gigatonnes (not the molecules, just carbon alone) that is cycled in a year. Yellow text is the natural carbon cycle, with red text showing human effects.[2] Notice that the 9 gigatonnes of carbon that humans are emitting (~35 gigatonnes of carbon dioxide) becomes an extra 4 gigatonnes in the atmosphere, an extra 3 gigatonnes of photosynthesis and an extra 2 gigatonnes in the ocean every year. This is how humans change the natural carbon cycle,” University of Calgary. (Image credits: https://energyeducation.ca/encyclopedia/Carbon_flux)
What is Stomatal Conductance?
Stomatal conductance (gs) is the plant process where stomata or tiny pores open or close in response to atmospheric conditions, soil, and plant water status. The process controls the movement of carbon dioxide (CO2) and water vapor between the atmosphere and the plants, and at a larger ecosystem scale, also terrestrial vegetation.
Higher land plants assimilate 15% of the atmospheric pool through photosynthesis annually, releasing the same amount through plant respiration, plant litter, and soil organic matter decomposition.
By regulating the concentrations of CO2 inside the leaf, stomatal conductance controls the amount of carbon fixed by photosynthesis to play a role in plant productivity. On larger scales, it influences the gross primary production (GPP) or total influx of carbon into the ecosystem.
The CO2 produced by plant respiration will also leave the plant through the stomata. So, stomatal conductance is pivotal in calculating net primary production (NPP), which is the gross carbon influx minus plant respiration or cost of plant growth and maintenance.
Annually, only 50 percent of carbon fixed by terrestrial photosynthesis is turned into biomass. The rest is lost due to plant respiration. The annual gross primary productivity by terrestrial ecosystems is 120 gigatonnes (GT) of carbon, and the amount lost through respiration is 60 GT, see Figure 1.
There are spatial variations in annual carbon sequestration depending on the vegetation. The evergreen forests fix 1000 g Cm-2, and the deserts only 30 g Cm-2. Due to carbon fertilization, the NPP of each ecosystem could be changing.
Stomatal conductance also influences plant water status, facilitates nutrient withdrawal from the soil, and drought sensitivity and tolerance. These functions also indirectly affect the carbon cycle.
Stomatal conductance changes due to several environmental and intrinsic plant factors (photosynthesis, transpiration, and signaling in the xylem). It can impact carbon flux to and from plants to influence the global carbon cycle.
The Affect of Environment on Stomatal Conductance
Given its various plant functions, scientific interest has increased in stomatal conductance and optimizing the process in current and future conditions of changing air temperature, CO2 levels, water deficit, drought, and ozone.
Scientific models that integrate the effects of environment and plant traits on stomatal conductance to estimate its contribution to the global carbon cycle are of great interest. Earlier models, which can be 40 years old, link stomatal conductance (gs) to environmental factors or net photosynthesis but do not consider drought effects. Models must demonstrate how stomata communicate with other cells in response to environmental factors, such as CO2 levels, air temperature, and air and soil moisture, to regulate stomatal conductance and optimize net primary productivity (NPP).
Rising CO2
There are varying estimates of the effect of rising CO2 on the carbon cycle, from none to significant.
As ambient carbon dioxide content increases, stomatal conductance decreases, reducing the entry of CO2 into the leaves and photosynthesis in the short term. In the long-term, stomata development is down-regulated, reducing gs in certain plant species. However, stomatal conductance can increase in specific CO2 ranges, up to a rise of ~50 ppm, the increase in greenhouse gases seen in the last three decades, leading to more photosynthesis. Though relatively rare, increases in gs can occur in woody taxa that are naturally adapted to warm low, humidity conditions.
Rising CO2 levels increase photosynthesis due to the higher gradient between intercellular and leaf surface CO2 and enhanced carboxylation rate of the enzyme RuBisCO in C3 plants. Over time, carbon fertilization can increase GPP. This increase was 6.2% between 1982–1999, with the Amazon Basin accounting for 33 percent of CO2 uptake. Globally, CO2 fertilization improved photosynthesis by 23 percent and was close to the 28 percent suppression of stomatal conductance by high CO2 levels.
Some scientists think rising CO2 levels do not affect respiration in the short term, but these do not represent long-term trends. Similarly, short-term or ten-year increases in photosynthesis due to carbon fertilization could taper off as plants get acclimatized. Most studies suggest that increasing CO2 levels will reduce respiration. One estimate suggests that the reduction can be 15-10 percent and improve carbon sequestration by 3·4 Gt of carbon each year.
Temperature
Rising temperatures will boost stomatal conductance to cool the plant through transpiration. The process increases by 40 percent in broadleaf and conifer species when temperatures increase by 10°C, from 30°C to 40°C. Stomatal conductance increases even if the plant water status is low and there is higher transpiration and intercellular CO2. More stomatal conductance due to higher temperatures can boost photosynthesis when enough soil water is present in the short term, but in the long term, this can deplete soil water.
As temperature increases, respiration rates will differ depending on the latitude. Ecosystem respiration doesn’t increase much in the tropics and the warm Mediterranean. Temperate forests have more respiration; most are seen in the cold boreal and tundra. In this case, tropics with more carbon fixation and lower respiration will sequester more carbon than cooler regions.
Ozone
Surface ozone ([O3]) concentrations have risen from 10 ppb before the industrial revolution to about 40 ppb over the northern temperate zone. Rises by another 50 percent in the current century can’t be ruled out. Ozone damages plants and reduces photosynthesis. Reduction in photosynthesis in crops, conifers, and broadleaved trees is linearly related to the rate of flux of O3 through stomata rather than the duration of exposure or dosage.
O3 decreases stomatal conductance by 13 percent and photosynthesis by 11 percent. However, angiosperms are affected more than conifers, and younger trees are more affected than older trees. Drought and elevated CO2 levels that reduce stomatal conductance protect the plants from O3 damage. Rises above current O3 levels will harm even the conifers. The ability of increases in O3 to depress photosynthesis is ignored in calculating carbon fluxes and should be considered, especially in the case of temperate and boreal forests.
Drought or Water deficit
Many species reduce their stomatal conductance when there is soil water, atmospheric vapor pressure deficit (VPD), and salinity. Nearly all species close their stomata during drought to prevent water vapor loss due to transpiration. The dehydrating roots send chemical signals through abscisic acid (ABA) and other compounds. Plants will keep their stomata closed during the day during water deficit conditions. However, this can interfere with their ability to reduce internal temperature, photosynthesis, and nutrient absorption, ultimately leading to their death. In some cases of extreme drought, plants have been observed to open their stomata to cool the plant, but the loss of water vapor will rapidly cause death. Lower levels of nitrogen and phosphorus increase the sensitivity of stomatal conductance to drought.
Lower soil moisture results in higher air temperature and increases respiration
Leaf Chlorophyll
The effect of leaf chlorophyll can be as crucial as water deficiency. Lower leaf chlorophyll levels can reduce stomatal conductance. Environmental factors have little seasonal variations. However, seasonal variations in chlorophyll concentration, larger in spring and autumn than in summer, will affect stomatal conductance and the carbon cycle between plants and the atmosphere.
Stomatal Conductance Across Ecosystems
Besides environmental and physiological factors, stomatal behavior differs among plant functional types according to their carbon cost of water use.
A study on stomatal conductance changes over the last century showed more reduction in grass-dominant ecosystems than dicot-rich communities. Moreover, soil nutrients or pH levels did not influence gs, except for low nitrogen levels.
Woody angiosperms with C3 photosynthesis across bioclimatic zones showed a strong convergence for species in the understorey 233 ± 86 mmol m-2 s-1. The mean stomatal conductance was also lower than woody species in open canopy habitats (266 ± 100 mmol m-2 s-1). This could result from the buffering provided by an over canopy from extremes of environmental conditions, an advantage the trees in open spaces didn’t have.
Modeling Stomatal Conductance

The effects of various factors on stomatal conductance are easier to outline separately. However, in nature, these can be acting together. Complex multi-factor models that integrate several or all relevant factors – environmental, plant physiology, ecosystem type, latitude, and plant type- can predict changes in stomatal conductance and its influence on NPP. Several such attempts then compare predictions with actual data collected on the site by instruments such as the CI-340-Handheld-Photosynthesis-System by CID Bio-Science Inc. A better understanding of stomatal conductance and its optimization can ensure food security and sustainable biomass sources for industries and natural ecosystems.
Sources
Johnston, A.S.A., Meade, A., Ardö, J., et al. (2021). Temperature thresholds of ecosystem respiration at a global scale. Nature Ecology & Evolution. DOI: 10.1038/s41559-021-01398-z
Allen, A. P., Gillooly, J. F., & Brown, J. H. (2005). Linking the global carbon cycle to individual metabolism. Functional Ecology, 19(2), 202-213. https://doi.org/10.1111/j.1365-2435.2005.00952.x
Baca Cabrera, J.C., Hirl, R.T., Schäufele, R. et al. Stomatal conductance limited the CO2 response of grassland in the last century. BMC Biol 19, 50 (2021). https://doi.org/10.1186/s12915-021-00988-4
Buckley, T. N. (2017). Modeling Stomatal Conductance. Plant Physiology, 174(2), 572-582. https://doi.org/10.1104/pp.16.01772
Carbon flux. Carbon flux – Energy Education. (n.d.). Retrieved April 7, 2023, from https://energyeducation.ca/encyclopedia/Carbon_flux
Cheng, D.-L., Li, T., Zhong, Q.-L., & Wang, G.-X. (2010). Scaling relationship between tree respiration rates and biomass. Biology Letters, 6(5), 715–717. https://doi.org/10.1098/rsbl.2010.0070
Engineer, C., Hashimoto-Sugimoto, M., Negi, J., Israelsson-Nordstrom, M., Azoulay-Shemer, T., Rappel, J., Iba, K., & Schroeder, J. (2016). CO2 sensing and CO2 regulation of stomatal conductance: Advances and open questions. Trends in plant science, 21(1), 16. https://doi.org/10.1016/j.tplants.2015.08.014
Friend, A., Arneth, A., Kiang, N., et al. (2007). FLUXNET and modelling the global carbon cycle. Global Change Biology, 13(3), 610–633. https://doi.org/10.1111/j.1365-2486.2006.01223.x
Gentine, P., Green, J. K., Guérin, M., Humphrey, V., Seneviratne, S. I., Zhang, Y., &; Zhou, S. (2019). Coupling between the terrestrial carbon and water cycles—a review. Environmental Research Letters, 14(8), 083003. https://doi.org/10.1088/1748-9326/ab22d6
Gifford, R. M. (2003). Plant respiration in productivity models: Conceptualisation, representation and issues for global terrestrial carbon-cycle research. Functional Plant Biology, 30(2), 171. https://doi.org/10.1071/fp02083
Houghton, R. (2003). The Contemporary Carbon Cycle. Treatise on Geochemistry, 473-513. https://doi.org/10.1016/B0-08-043751-6/08168-8
Lin, Y., Medlyn, B. E., Duursma, R. A., et al. (2015). Optimal stomatal behaviour around the world. Nature Climate Change, 5(5), 459-464. https://doi.org/10.1038/nclimate2550
Marchin, R. M., Backes, D., Ossola, A., Leishman, M. R., Tjoelker, M. G., & Ellsworth, D. S. (2021). Extreme heat increases stomatal conductance and drought‐induced mortality risk in vulnerable plant species. Global Change Biology, 28(3), 1133–1146. https://doi.org/10.1111/gcb.15976
Matsumoto, K., Ohta, T., & Tanaka, T. (2005). Dependence of stomatal conductance on leaf chlorophyll concentration and meteorological variables. Agricultural and Forest Meteorology, 132(1-2), 44–57. https://doi.org/10.1016/j.agrformet.2005.07.001
MEDLYN, B., DUURSMA, R., EAMUS, D., ELLSWORTH, D., PRENTICE, I., BARTON, C., CROUS, K., DE ANGELIS, P., FREEMAN, M., & WINGATE, L. (2011). Reconciling the optimal and empirical approaches to modelling stomatal conductance. Global Change Biology, 17(6), 2134–2144. https://doi.org/10.1111/j.1365-2486.2010.02375.x
Murray, M., Soh, W. K., Yiotis, C., et al. (2019). Convergence in Maximum Stomatal Conductance of C3 Woody Angiosperms in Natural Ecosystems Across Bioclimatic Zones. Front. Plant Sci., 10, 558. https://doi.org/10.3389/fpls.2019.00558
Perez-Martin, A., Flexas, J., Ribas-Carbó, M., Bota, J., Tomàs, M., Infante, J. M., et al. (2009). Interactive effects of soil water deficit and air vapour pressure deficit on mesophyll conductance to CO2 in Vitis vinifera and Olea europaea. J. Exp. Bot. 60, 2391–2405. doi: 10.1093/jxb/erp145
Ram, H., Kaur, A., Gandass, N., et al. (2020). Stomatal Adaptive Response in Plants Under Drought Stress. Eds Kuila, A. Plant Stress Biology, Apple Academic Press. https://doi.org/10.1201/9781003055358
Running, S.W., Thornton, P.E., Nemani, R., Glassy, J.M. (2000). Global Terrestrial Gross and Net Primary Productivity from the Earth Observing System. In: Sala, O.E., Jackson, R.B., Mooney, H.A., Howarth, R.W. (eds) Methods in Ecosystem Science. Springer, New York, NY. https://doi.org/10.1007/978-1-4612-1224-9_4
Urban, J., Ingwers, M., McGuire, M. A., & Teskey, R. O. (2017). Stomatal conductance increases with rising temperature. Plant signaling & behavior, 12(8), e1356534. https://doi.org/10.1080/15592324.2017.1356534
Wittig, V., Ainsworth, E., & Long, S. (2007). To what extent do current and projected increases in surface ozone affect photosynthesis and stomatal conductance of trees? A meta-analytic review of the last 3 decades of experiments. Plant, Cell & Environment, 30(9), 1150–1162. https://doi.org/10.1111/j.1365-3040.2007.01717.x
Zhang, X., Zhang, Y., Tian, J., Ma, N., & Wang, Y.-P. (2022). Co2 fertilization is spatially distinct from stomatal conductance reduction in controlling ecosystem water-use efficiency increase. Environmental Research Letters, 17(5), 054048. https://doi.org/10.1088/1748-9326/ac6c9c
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






