January 14, 2021 at 9:59 am | Updated March 15, 2022 at 11:47 am | 7 min read
Fine root turnover is important for the nutrient acquisition of trees and in nutrient cycling for the entire ecosystem. Hence, it is important to understand how elevated levels of atmospheric carbon dioxide are influencing root turnover in high altitude forests. Special tools, suited to non-destructive and repeated data collection, are crucial to the success of the time-series experiments needed for this kind of research.
Does Increased Carbon Dioxide Affect Underground Growth?
The unprecedented increase in atmospheric carbon has led to an increase in photosynthetic activity. Scientists calculate an average increase in photosynthesis by 60% in trees.
How each species responds to elevated carbon levels depends on various other factors, such as crown architecture, availability of nitrogen, season, and presence of pollutants.
Subscribe to receive our monthly round-up of articles.
It is not clear if an increase in photosynthesis translates into more root growth, however. Nearly half of a plant’s biomass lies in roots, which are classified as being coarse or fine. Fine roots make up less than 2% of the total root biomass. However, because they are produced each year and have a high turnover, they contribute to 33-67% of the net primary productivity (NPP) in forests.
Fine root production (FRP) and mortality (FRM) are important for an ecosystem, as trees access nutrients and water through them. After their death, they add carbon and humus to the soil, making them a vital part of nutrient cycling.
Scientists have found that, in some cases, more carbon is allocated to produce roots; however, the evidence for this is not conclusive. Moreover, the effect of altitude on changes in photosynthesis is also not established.
White birch (Betula platyphylla var. japonica) grows in a wide range of environmental conditions and is dominant in many forest types.
Japanese scientists—Wang, Fujita, Nakaji, Watanabe, Satoh & Koike—wanted to see how elevated carbon dioxide (CO2) would affect birch root turnover in volcanic ash soils that are poor in both phosphorus and nitrogen.
Challenge: Finding a Non-destructive Continuous Soil Sampling Method
The growth of white birch in volcanic soils (VA) was compared to white birch in fertile brown forest soils (BF).
The split-plot experiment was conducted in two FACE systems, where two levels of CO2 were provided: one had the current atmospheric conditions (380-390 µmol mol-1 CO2) and the second an elevated level (500 µmol mol-1 CO2). Soil type was the second level in each FACE. Three replicates were used in the trial.
As root turnover was the central focus of the experiment, the scientists needed to find a non-destructive method that could be used to study four randomly selected tree seedlings in each FACE over three years.
Besides looking for a method that was non-destructive, the scientists wanted a method that could monitor the same roots at intervals lasting over years.
The root ingrowth method uses buried mesh bags of soil, allowing roots to grow within them. To study root growth, the root bag must be removed before the death of the roots inside.
There are several reasons why the root ingrowth method was not suitable for the present experiment. First, the scientists would have needed prior knowledge of root longevity that they did not have. Second, since the roots are destroyed prematurely, the potential full length of the roots would remain unknown. Finally, the injury to the root tissue, as well as disturbance to the soil, can destroy other fine roots of the plant and influence further growth.
While the ingrowth method can underestimate fine root turnover, sequential coring can overestimate the parameters.
Sequential coring involves taking soil cores from different depths and various spots and homogenizing different samples taken at the same depth. The root parameters of interest are manually measured, and the whole sampling requires a lot of time. While processing, there is a danger that living roots can dry up and be counted as dead roots. Also, sequential coring is neither suitable for a time-series experiment, nor when small-scale variability is important.
Hence, the scientists ruled out both ingrowth core and sequential coring for their experiment.
Solution: CI-600 Root Scanner
Instead, the scientists decided to use a minirhizotron. They selected the CI-600 In-Situ Root Imager.
In 2010, at the same time that they transplanted three-year-old white birch saplings into the experimental plots, they installed transparent acrylic tubes about 0.5 meters long into the soil. The root tubes were 5.08 cm wide and inserted into the soil at an angle of 45° to the soil surface. As the saplings established themselves, the roots grew undisturbed around the root-tubes.
The researchers inserted the CI-600 root scanner into each tube and captured digital images of roots at depths of 0-15 cm and 15-30 cm. In the shallow soil of 0-15 cm, the scientist found many roots belonging to grasses. Hence, for accurate measurements, they relied on the scans made at the deeper soil layers of 15-30 cm, where only tree roots were found.
In total, the scientists installed 24 root tubes, with 12 in each FACE system.
Since fine roots need one year to grow and establish after transplantation, the scientists refrained from collecting data during this time. In the second year, they scanned the roots once every three weeks for three years, except during snow.
The scientists used the program WinRHIZOTron from Regent Instruments to analyze the captured images. They classified the roots using the following method:
- White roots as new
- Roots remaining white or turning brownish were live
- Black or wrinkled roots as dead
The lengths and diameters captured in images were traced and measured. The sum of the lengths of new roots and an increase in the length of older roots was considered to be fine root production. Root length that was no longer visible was considered to be fine root mortality.
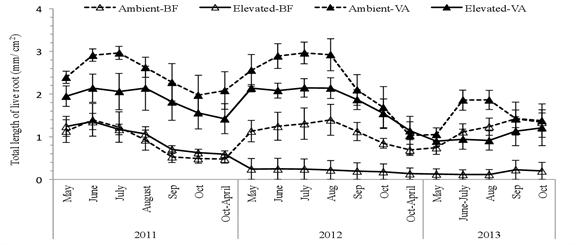
Figure 1: Relative length of live fine root (standing crop) of white birch saplings growing under ambient and elevated CO2 levels in volcanic soils (VA) and brown forest soils (BF), Wang et al. 2016. (Image credits: https://doi.org/10.1007/s00468-015-1282-4)
Benefits of Using the CI-600 for Root Imaging
The scientists state that they chose the minirhizotron, manufactured by CID Bio-Science Inc., because it was superior to the other methods. They could identify the same roots in scans on successive dates, as the method is non-destructive. They could quantify all the parameters important for the experiment, such as root length, density, diameter, mortality, and longevity.
Moreover, they were able to monitor growth in 24 trees with the help of one scanner. They collected data around 15 times each year, so they had hundreds of data scans for their analyses.
The scans gave high-resolution images up to 23.5 million pixels. The scanning resolutions were also high and ranged from 100-600 DPI. It is possible to turn the camera, located at the end of the imager, around 360 degrees to get a scan of all the roots around the root tube at predetermined soil depths.
The CI-600 is light and convenient to carry, and the scientists reported that it fit well with the diameter of the root tubes they had used. The software with the imager saves the scans for transfer to a computer through a USB cable.
Elevated Carbon Dioxide Has Varying Effects over Time
In the first year, an increase in CO2 did not result in any difference in the length of the fine root in BF, but in the second year, the fine root length was reduced. However, in VA soils, elevated CO2 levels lowered fine root length in all three years, as shown in Figure 1.
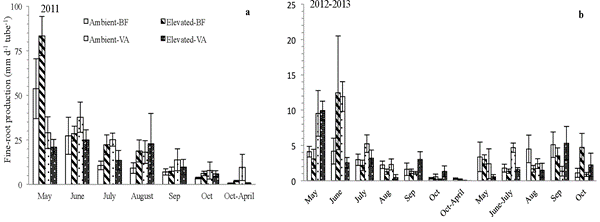
Figure 2: “Fine root length production rate of birch seedlings growing under elevated and ambient [CO2] on volcanic ash (VA) and brown forest (BF) 709 soil. Each value is the mean of six replications, the vertical bar in the column denotes SE. The letter a and b denote the graph of different years,” Wang et al. 2016. (Image credits: https://doi.org/10.1007/s00468-015-1282-4)
In both soils, there was less turnover of fine roots in the first and second year under elevated CO2 compared to ambient CO2 levels. This was because fine root mortality was lower, and the increase in total root length was less due to soil conditions; see Figures 2 and 3.
When there is more CO2, the trees increase water use and nutrition absorption to invest in above-ground growth. After the above-ground portion is established in the new tree, it diverts its resources to build roots in the third year.
Fine root longevity increased, especially in VA, in the first two years due to increased CO2 levels, indicating that soil nutrition is also important. When there is less nutrition, trees allocate fewer resources to grow new roots and retain older growth, especially if the costs of producing new roots are higher than maintaining old roots.
In the third year, there was a high degree of turnover in fine roots, suggesting increased production and death of fine roots; see Figure 3.
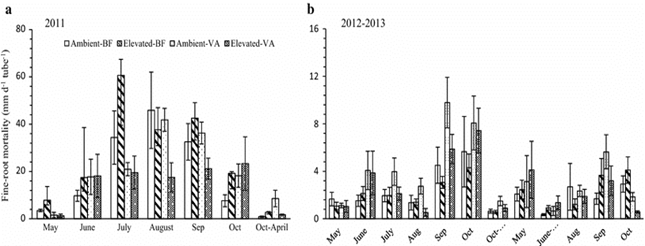
Figure 3: “Fine root length mortality rate of birch seedlings growing under elevated and ambient [CO2] on volcanic ash (VA) and brown forest (BF) soil. Each value is the mean of six replications; the vertical bar in the column denotes SE. The letters a and b denote the graph of different years,” Wang et al. 2016. (Image credits: https://doi.org/10.1007/s00468-015-1282-4)
The scientists think that mycorrhizal growth could also have influenced fine root lifespan. With an increase in symbiosis, there is less nitrogen accumulation, which results in a longer root lifespan.
So, mycorrhizal growth seems to increase under elevated CO2 levels in the initial years, which helps new plants establish.
The higher turnover in the third year can also lead to high carbon sequestration in soils.
Importance of Multi-season Experiments
Short experiments provide only limited information. To get a better picture of dynamics in long-lived trees, longer experiments are more suitable. For these experiments, non-destructive and innovative instruments, like the CI-600 In-Situ Root Imager, are vital. By collecting data of difficult underground processes for three years, the scientists were able to gather valuable information on carbon flux in white birch forests, which can help in the adaptation and mitigation of climate change.
Tools: |
See More: |
| Cassava Cultivation and the Photosynthesis Process in Drought
Measuring How Microclimate Effects Turmeric Yield Rising CO2, Crop Yield, and Leaf Area Measurement |
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Feature image courtesy of Quillshadow
Sources
Hertel, D., Leuschner, C. (2002). A comparison of four different fine root production estimates with ecosystem carbon balance data in a Fagus–Quercus mixed forest. Plant and Soil 239, 237–251. https://doi.org/10.1023/A:1015030320845
Schroth, G., Kolbe, D. (1994). A method of processing soil core samples for root studies by subsampling. Biol Fertil Soils 18, 60–62. https://doi.org/10.1007/BF00336446
Steingrobe, B., Schmid, H., & Claassen, N. (2001). The use of the ingrowth core method for measuring root production of arable crops – influence of soil and root disturbance during installation of the bags on root ingrowth into the cores. European Journal of Agronomy, 15 (2), 143-15. https://doi.org/10.1016/S1161-0301(01)00100-9.
Wang, X., Fujita, S., Nakaji, T. et al. (2016). Fine root turnover of Japanese white birch (Betula platyphylla var. japonica) grown under elevated CO2 in northern Japan. Trees 30, 363–374. https://doi.org/10.1007/s00468-015-1282-4
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






