October 21, 2020 at 10:03 pm | Updated April 18, 2022 at 1:17 pm | 7 min read
There is no denying the importance of light for the growth of plants; however, there are many other environmental factors—such as temperature, moisture and nutrient availability, and soil type–that also influence plant growth. These factors do not act in isolation and are, many times, correlated. So, it is interesting to see how light works in combination with other resources to influence plant growth in the wild or in farms. Accurate and quick measurements are vital to advance the studies in this area of research.
Interactions of Light and Other Environmental Factors
Light is one of the essential factors for photosynthesis by which plants produce their food. Plants use light energy to combine the water drawn in from roots with carbon dioxide taken in through the leaf stomata to produce simple sugars. By combining the products of photosynthesis with minerals, which are also absorbed through roots, plants produce all the other compounds they need like proteins, fats, pigments, fibers, etc.
The portion of visible light spectrum between 400-700 nm is vital for photosynthesis and is called the photosynthetically active radiation. When there is more solar radiation, ambient and soil temperature will rise. This, in turn, can increase evaporation of moisture from soils, decreasing their availability for plants. In the plants themselves, the temperature can influence many physiological processes.
Subscribe to receive our monthly round-up of articles.
Moreover, in response to light, stomata open during the day to let in carbon dioxide for photosynthesis. However, this also results in more transpiration, which encourages movement of water loaded with nutrients from the roots through the plants. Stomata close at night or when there is drought stress.
Soil fertility and structure also influence how much water and nutrients are available for plants.
Thus, we see that the different environmental factors influence each other, and their interactions will also affect plant physiology, growth, and yield.
Hence, researchers try to observe the work of as many factors together as possible to get a more nuanced picture of what is happening in plants. It is easier to design experiments and study each factor separately, but they can’t capture the complexity of the real conditions on the field under which plants grow.
Plant Measurement Instruments
Scientists need reliable, accurate instruments that they can carry to the field for research. Often, plant scientists are faced with the problem that their study area, which could be farms, forests, or greenhouses, are far from any laboratory. Making frequent trips from the field to laboratories are either difficult or very expensive. Portable scientific devices, which can make spot measurements, are the answer to this problem.
In the following sections, we will discuss how different instruments help in studying the effect of environment on plants.
Light and Temperature
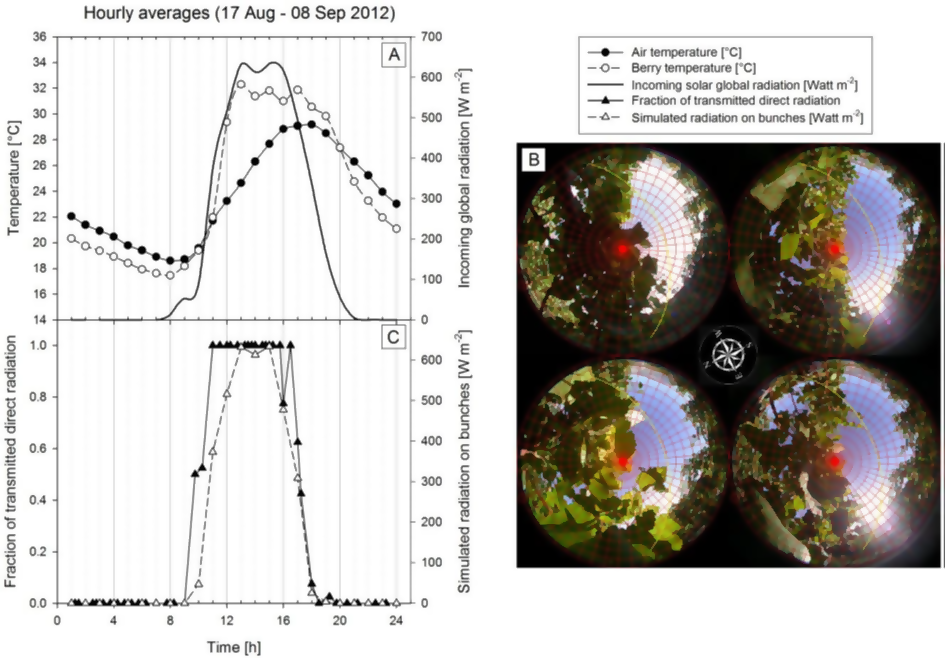
Figure 1: “A: The graph shows the effect (averaged over three weeks and four South exposed bunches) of the solar radiation (plain thick line) on bunch temperature (open circles) as compared to air temperature (closed circles). A 10 °C increase of berry temperature was observed as a result of exposure to direct light. B: Registered hemispherical images collected close to the same four bunches used for the hourly reconstruction of the percentage of direct radiation with GLA 2.0. In the images, the 7.5° sky regions and the sun path of a specific day are shown. C: Results of the hourly reconstruction from hemispherical pictures. On average, 78% of the direct radiation resulted available on the four bunches, particularly during the central and warmer part of the day. Only early in the morning and late in the evening there is a masking effect due to the canopy.” (Image credits: https://www.researchgate.net/publication/264541831_Hourly_simulation_of_grape_bunch_
light_microclimate_using_hemispherical_photography)
In Italy, scientists wanted to find how much light grape bunches received, as this affected their temperature and composition.
They decided to take an indirect approach, since it is not easy to measure light reaching grape bunches throughout the day. They measured the gaps in the canopy of the grape plants and the light that was coming through them, with the help of hemispherical pictures, to help find out how the slope, shading effects of surrounding hills, trellis orientation and height, and position of grape bunches on the vine affected light availability for the fruit bunches.
Among the different instruments used on the field was the CI-110 Digital Plant Canopy Imager by CID Bio-Science to take hemispherical photos over four bunches of grapes to show the gaps in the canopy, effective leaf area index (LAI), and under-canopy light; see Figure 1B for hemispherical images.
The CI-110 Digital Plant Canopy Imager has a long probe with a camera fitted at the end to take 1500 fisheye images of the canopy and calculate LAI, using the Gap Fraction Method. There are also 24 sensors on the probe arm to measure PAR light. By combining the two sets of data, many canopy parameters are calculated.
The experiment lasted a year, during which time continuous data was recorded and averaged every 15 minutes.
Scientists found that row orientation affects grape light availability. The hemispherical pictures showed that seventy-eight percent of the sun’s light reached the grape bunches and maximum light reached the grape bunches during mid-day. There was shading only during early morning and late evening. This was reflected in fruit temperatures that were highest around 1 pm and were 7.70C higher than air temperatures, while the grape bunch temperatures were lower by 1.50c than air temperature in the early morning and evenings.
Light and Nutrients
It is important to understand how plants adapt to their environment and whether plants can respond to different environments by changing growth parameters to survive in them. This ability to change growth parameters is called phenotypic plasticity. Plants that multiply through vegetative means, for example with rhizomes, are usually thought to have less flexibility to adjust to new environments compared to plants that propagate through seeds.
A study in China used the stoloniferous herb, Glechoma longituba L., that is widespread in forests and grasslands. The scientists wanted to see how vegetative propagules of this herb, a close relative of ground ivy, would respond to different light intensities and nutrient availability.
They checked the leaf area, root length, biomass, number of ramets (propagules/offshoots that can develop into a new plant), and weight of ramets. To measure the leaf area, scientists used the CI-203 Handheld Laser Leaf Area Meter, manufactured by CID Bio-Science.
The CI-203 Handheld Laser Leaf Area Meter is a portable laser scanner that can be operated with a single hand. It makes rapid, non-destructive measurements of leaf length, width, area, perimeter, shape factor, and aspect ratio. The built-in GPS tags location to each data point, so that time-series measurements are also possible. A data-logger can store information for later transfer to computer through a USB connection.
The scientists found that when the light intensity was greater, the plants showed an overall better growth (there were more ramets produced and the biomass increased), while the plants in areas with low light intensity and high soil nutrients showed the same growth features as the mother plants.
The root-shoot ratio, which is a ratio of the size of the root-system to above ground vegetation parts, was varied to improve the use of light and nutrients in the two environments. In each case, the herb was able to survive by adjusting stem, leaf, and root size to the different environments.
Light and Soil Moisture
Light intensity and soil moisture are important for plant growth. In natural conditions, these factors also affect the distribution of plants. Both light intensity and soil moisture can influence photosynthesis, transpiration, and water and nutrient uptake in plants
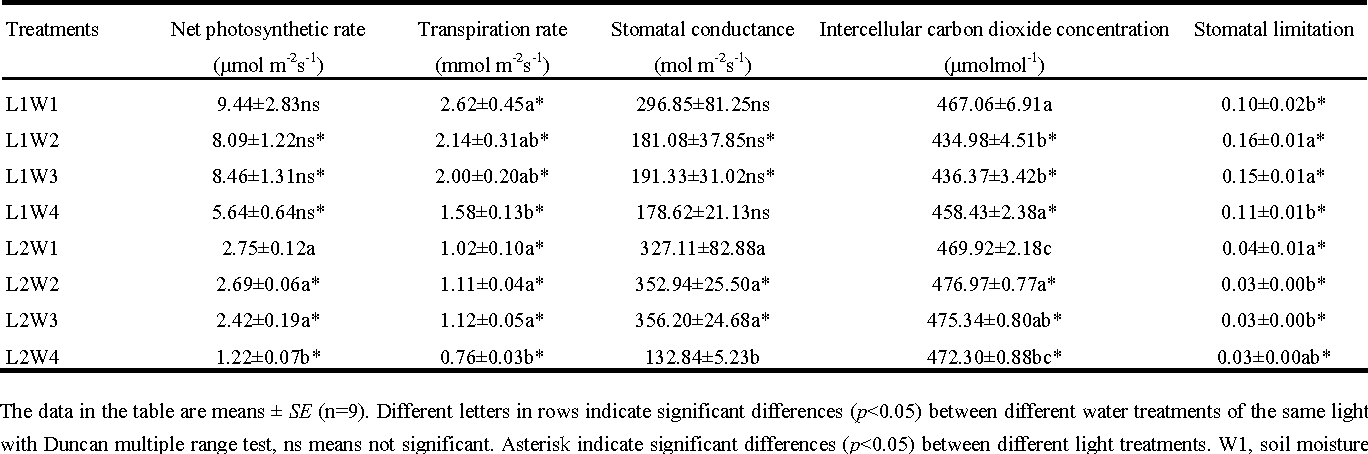
Figure 2: W1, soil moisture level at 80%−75%, W2 at 60%−55%, W3 at 40%−35%, W4 at 20%−15%; L1, full light in the greenhouse, L2, shading light in the greenhouse. (Image credits: https://doi.org/10.1007/s11676-013-0352-y)
The deciduous shrub Amorpha fruticosa is common in warm temperate regions in China. A greenhouse experiment was conducted to see how the plant performed under different water and light conditions.
Plant biomass, height, basal diameter, and crown area were recorded every ten days. These were compared to the amount of photosynthetic pigment and the water content in the soil. The CI-340 Handheld Photosynthesis System by CID Bio-Science recorded photosynthetic parameters, such as net photosynthetic rate, stomatal conductance, transpiration rate, and carbon dioxide concentration.
The CI-340 Handheld Photosynthesis System is a portable tool designed for field use to make non-destructive measurements. It is a gas analyzer that calculates photosynthesis and transpiration by measuring the concentrations of different gases before and after they enter the leaf chamber. Stomata conductance is calculated based on transpiration. The CI-340 has modules that can control carbon dioxide, water vapor, temperature, light intensity, and chlorophyll fluorescence to manipulate conditions for experiments. It is suitable for use with open and closed systems.
When soil moisture was less, the plants showed a decrease in growth rate, size, and photosynthetic parameters. The plants also grew more roots. When light intensity was less, photosynthetic parameters, water-use efficiency, and biomass accumulation declined. In this case, the plant allotted more of its resources to stem and leaf growth. The ideal growing conditions for the shrub was seventy-five to eighty percent of soil water holding capacity and one hundred percent of light intensity in the greenhouses. See the first row in Figure 2 to check the best results in the experiment.
Tools for Every Task
The needs of scientists for spot measurements on the field are receiving increased attention. More tools are being developed so that scientists can make accurate recordings of even complex physiological processes in the fields. In a world with a changing climate, researchers must be able to track responses of wild plants and crops. It can provide information to help in adaptation and mitigation of climate change and to optimize crop growth and yields.
—
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Featured blog image courtesy of David Tyler
Sources
Chapter 6, Taiz and Zeiger’s Plan Physiology. (2006, March 6). Botany 4400/5400, Lecture 22. Retrieved from http://www.uwyo.edu/botany4400/lecture%2022.htm
Chu, Y., Yu, F.H., & Dong, M. (2006). Clonal Plasticity in Response to Reciprocal Patchiness of Light and Nutrients in the Stoloniferous Herb Glechoma longituba L. Journal of Integrative Plant Biology, 48: 400-408. doi:https://doi.org/10.1111/j.1744-7909.2006.00237.x
What is Photosynthetically Active Radiation? (2010, August 12). Retrieved from https://www.fondriest.com/news/category/oceans-coasts.
Zhang, X., Tan, X., Wang, R. et al. (2013). Effects of soil moisture and light intensity on ecophysiological characteristics of Amorpha fruticosa seedlings. Journal of Forestry Research, 24: 293. https://doi.org/10.1007/s11676-013-0352-y
Zorer, R., Moffat, T., Strever, A., & Hunter, J.J. (2013). Hourly simulation of grape bunch light microclimate using hemispherical photography. Ciência e Técnica Vitivinícola, 28:1031 – 1034. Retrieved from https://www.researchgate.net/publication/264541831_Hourly_simulation_of_grape_bunch_light_microclimate_using_hemispherical_photography
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






