October 21, 2020 at 10:03 pm | Updated March 14, 2022 at 1:06 pm | 7 min read
Salinity inhibits growth and development in crops. Due to the extent of damage salinity causes to agriculture, it is identified as a major threat to food production. Therefore, practices to deal with the effects of salinity on crops are receiving a lot of attention. To be able to provide proper recommendations that are also sustainable, it is first necessary to find out how salinity affects plants in different stages of their growth. Precise instrumentation plays a major role in monitoring this stress during research on the farm.
Salinity Stress in Crops
An estimated 800 million hectares are affected globally by salinity, according to FAO.
Salinity stress is due to the presence of high amounts of salt or sodium chloride (NaCl) in the soil water or irrigation waters.
Subscribe to receive our monthly round-up of articles.
The extent to which a plant is affected by salinity depends primarily on the species. There is a threshold level of salt concentration that each plant species can tolerate. When the salt content in the soil increases beyond this level, the plant begins to suffer.
Salinity is measured as the electrical conductivity (EC) of soil solution. Most crops can withstand salinity of 1 to 1O dS/m. Soil is said to be saline when the EC is 4 dS/m and that it has 40 millimolars (mM) of NaCl.
Crops such as sugar beet, wheat, rye, barley, and cotton are considered to be salt tolerant, yet even their growth will be affected if salinity is more than 1O dS/m
Cereal crops are the most susceptible to salt stress, with effects seen in vegetative and early reproductive phases.
Other Factors
Soil attributes, water, and climatic conditions also affect plants’ ability to tolerate salinity stress. Crops, especially in arid and semi-arid regions, are impacted by salinity. As more and more farms get irritated, the risk of salinity also rises.
Effects of Salinity
Salinity affects plants in different ways from the beginning of its life cycle, when it is just a seedling, to yield production. This stress has both acute and chronic effects on plants.
Growth response
Early symptoms manifest within a period of hours to days. One of the first processes to be affected is osmotic regulation, which causes water stress. There is also increased uptake of sodium (Na+) and chloride (Cl−) ions, leading to nutritional imbalance. In response, the stomata close and reduce photosynthetic rates and cell division.
In the long term, the ion levels in the plants build up. As a result, many vital metabolic processes are affected, including
- Photosynthesis, by changing photosynthesis electron transport, chlorophyll content, and fluorescence
- Water-cycle
- Levels of reactive oxygen species generation at the cellular level
- Antioxidant defences
- Root to canopy ratio
Yield response
Salinity affects yield in crops gradually. Initially, when the concentration of salts is low, reduction is zero or negligible. As salinity stress increases, yield decreases in inverse proportion. In many cases, if salinity is severe, the plant can die before they fruit or produce grains, or the yield is so less that it is uneconomical. If salinity exceeds the threshold level, the yields of crops are definitely affected.
Research has focused on salinity stress in different crops. Keeping the effects in mind, varieties are developed following various strategies to improve crop tolerance of salinity. We will show how salinity stress was detected in different crops.
Varietal Response to Salinity
Wheat (Triticum aestivum L.) is considered to be more tolerant of salinity stress but is still affected by salt stress. A study delved deeper to find if there was a variation in salt tolerance in different varieties. These varieties were produced by incorporating a chromosome from a wild wheat relative. Scientists tested the eight new varieties and the wild parent wheat under control conditions with 1.5 dS/m and salinity of 15 dS/m. The nine varieties were tested for short- and long-term responses such as
- transpiration rate,
- stomatal conductance,
- net carbon dioxide (CO2) assimilation rate,
- CO2 fixation,
- green biomass production,
- and grain yield per plant.
These processes are affected by salt stress due to osmotic inhibition, where the plants cannot absorb water, or due to increased absorption of salt ions. Salinity was measured in the irrigation water supplied to the eight varieties. Plant physiology was monitored by recording CO2, stomatal conductance, transpiration rate, assimilation (fixed CO2), and net photosynthesis. Recordings were made in the forenoon, non-destructively, using fully expanded leaves, with the help of the CI-340 Handheld Photosynthesis System, produced by CID Bio-Science.
The CI 340 is a small, portable, handheld device that uses infrared light to measures gas concentrations entering and leaving the leaf chamber. The leaf chamber encompasses the whole or part of a leaf to estimate transpiration, stomatal conductance, and CO2 absorption.
- The instrument measures water vapor entering the leaf tissue to record transpiration.
- CO2 entry and amount of water vapour given out by the leaf is the measure for stomatal conductance.
- The rate of photosynthesis is calculated by measuring the amount of CO2 in the leaf chamber, before the gas enters the leaf and after it exits it. The difference shows the amount of CO2 the leaf has taken up or assimilated for photosynthesis.
The advantage of this method is that the leaf in question is not destroyed and can be tested several times again.
To estimate growth and yield, plant weight (fresh and dry), number of tillers and leaves, spike parameters, grains/spike, grain weight, and plant yield were also recorded.
All the varieties performed better in non-saline conditions. There was less transpiration, stomatal conductance, and CO2 assimilation under saline conditions. This led to less green biomass accumulation and grain yield. There was a significant difference in how the eight varieties responded to salinity in all the parameters measured, as can be seen in the Figures 1-3.
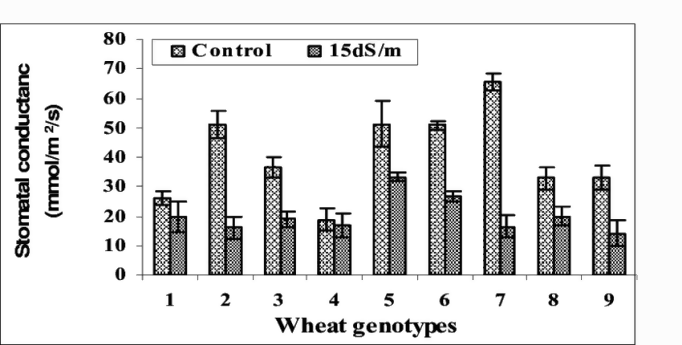
Figure 1: “Effect of different salinity levels (1.5 and 15 dS/m) on stomatal (c) conductance of wheat lines.” Afzaal et al. 2006 (Image credits: https://www.researchgate.net/publication/ 268284307_Differences_in_agronomic_and_physiological_performance_of_various_wheat_genotypes_grown_under_saline_conditions)
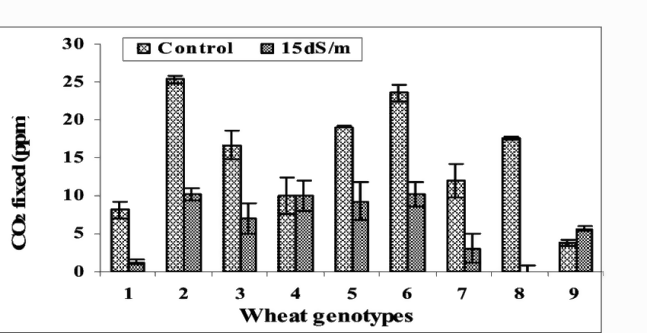
Figure 2: “The effect of different salinity levels (1.5 and 15 dS/m) on carbon dioxide fixation in lines of wheat (1-8) and parent Chinese Spring (9).” Afzaal et al. 2006 (Image credits: https://www.researchgate.net/publication/ 268284307_Differences_in_agronomic_and_physiological_performance_of_various_wheat_genotypes_grown_under_saline_conditions)
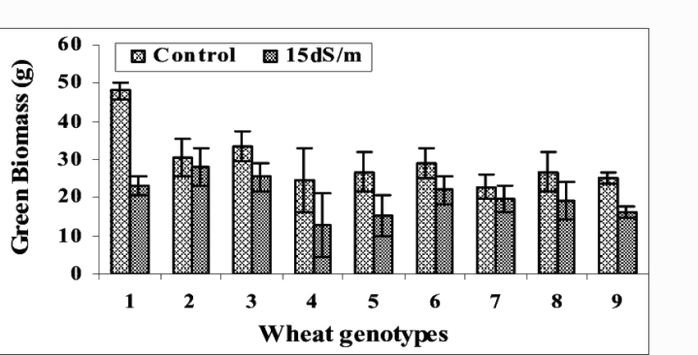
Figure 3: “Effect of different salt levels (1.5 and 15 dS/m) on green biomass (g) of lines of wheat.” Afzaal et al. 2006 (Image credits: https://www.researchgate.net/publication/ 268284307_Differences_in_agronomic_and_physiological_performance_of_various_wheat_genotypes_grown_under_saline_conditions)
The response of Salinity –Sensitive Crops
Tomato (Solanum lycopersicum) is moderately sensitive to salinity. An important cash crop, it needs frequent irrigation, as it is grown worldwide even in places with less rainfall and is, therefore, prone to salinity. In tomatoes, salinity restricts the ability of the plants to absorb water from the soil and all the nutrients that are usually dissolved in it. As a result, the yield is reduced.
When salinity increases in the leaf tissue, the stomata close. As a result, there is no transpiration, so less water is withdrawn from the soil. Moreover, CO2 entry, which takes place through stomata, is lessened. Salt stress can also break down chlorophyll, which captures light needed for photosynthesis. Thus, all three requirements for photosynthesis are severely limited by salinity, affecting the plants’ ability to manufacture food that fuels plant growth and fruit production. If the short-term effects of these processes are determined, steps can be taken by suitable agricultural practises that limit long-term damages to the crop yield.
An experiment tried to detect salt stress in plants at -0.2,-0.4,-0.6,-0.8 and -1.0MPa of reduced osmotic potential due to salinity. Once again, this study measured rates of photosynthesis, transpiration, and stomatal conductance with the help of the CI-340 Handheld Photosynthesis System, produced by CID Bio-Science. Also, amounts of chlorophyll and the efficiency of Photosystem II in chlorophyll in absorbing light were measured.
The adverse effects of salinity could be seen on all the physiological parameters after 14 and 21 days from sowing, even at the least osmotic potential of -0.2 MPa caused due to salinity.
Leaf Area Measurements
Besides measuring the physiological processes, it is also possible to detect salt stress by studying its effects on growth parameters with sophisticated devices. This was the approach taken to see how different levels of salinity caused by 0, 25, 50, 100 and 150 mM of NaCl affected the vegetative growth of karkade (Hibiscus sabdariffa var. sabdariffa). The number of germinated seedlings and delay in emergence were recorded. Also, length of seedlings and area of leaves were measured using the CI-202 Portable Laser Leaf Area Meter.
The CI-202 Leaf Area Meter is a portable device that smoothens the leaves and has a sliding high- resolution laser scanner that moves over the leaves to measure its length, width area, and shape using pre-programmed formulae. The method is non-destructive and gives rapid readings. The device can store around 8000 readings in its built-in logger.
The karbade seedlings were able to tolerate lower levels of salinity. However, at higher salinities of 100 and 150 mM, the leaf area, as well as the length of seedlings was reduced. This occurs because the osmotic potential in seedlings decreases as salinity increases.
Instruments for Research and Farm
Using small but highly advanced tools, scientists can easily track changes in crops with field measurements that are quick and do not require lengthy laboratory procedures. The devices can be applied to study a wide range of physiological processes and vegetative parameters. With the help of these devices, researchers have gained valuable insights into the effects of salinity at various stages of grains, pulses, fruits, and vegetables to advise farmers.
—
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Sources
Abdelnasser, G. (2017). Physico-chemical changes in karkade (Hibiscus sabdariffa L.) seedlings responding to salt stress. Acta Biologica Hungarica, 68. DOI: https://doi.org/10.1556/018.68.2017.1.7
Acosta-Motos J., Ortuño M., Bernal-Vicente A., Diaz-Vivancos P., Sanchez-Blanco M., Hernandez J. Plant Responses to Salt Stress: Adaptive Mechanisms. Agronomy. 2017;7:18. doi: 10.3390/agronomy7010018.
Afzaal, M., Farooq, S., Akram, M., Naz, F., Arshad, R., & Bano, A. (2006). Differences in agronomic and physiological performance of various wheat genotypes grown under saline conditions. Pakistan Journal of Botany. 38:1745-1750. Retrieved from https://www.researchgate.net/publication/268284307_Differences_in_agronomic_and_physiological_performance_of_various_wheat_genotypes_grown_under_saline_conditions.
Jadav, S., Bardhan, K., & Patel, D. (2017). Differential Early Stage Water Relations, Gaseous Exchange and Photosynthetic Responses of Tomato (Solanum lycopersicum L.) to Water and Salt Stress. British Journal of Applied Science & Technology. 19. 1-13. DOI: 10.9734/BJAST/2017/32201
Hernández J. A. (2019). Salinity Tolerance in Plants: Trends and Perspectives. International journal of molecular sciences, 20(10), 2408. doi:10.3390/ijms20102408
Isayenkov, S. V., & Maathuis, F. (2019). Plant Salinity Stress: Many Unanswered Questions Remain. Frontiers in plant science, 10, 80. doi:10.3389/fpls.2019.00080
Maas E.V. (1993) Plant growth response to salt stress. In: Lieth H., Al Masoom A.A. (eds) Towards the rational use of high salinity tolerant plants. Tasks for vegetation science, vol 27. Springer, Dordrecht. DOI:https://doi.org/10.1007/978-94-011-1858-3_31
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






