March 7, 2024 at 5:32 pm | Updated April 15, 2026 at 2:30 am | 6 min read
- Root turnover rates are complex to measure as growth and death co-occur in plants.
- Minirhizotron is a standard method for estimating root turnover as it is non-destructive, rapid, and accurate.
- One challenge in the widespread use of minirhizotrons for root turnover is the lack of analysis methods to directly convert root scans to turnover estimation.
Root turnover contributes to nutrient cycling, soil carbon storage, and ecosystem functions. It is crucial for crops and natural habitats. However, the measurement of root turnover rates is uncertain as they are methodologically challenging, and roots exist underground, making data collection difficult. This article will cover minirhizotron applications for non-destructive, efficient, and quantitative measurement to address scientists’ challenges in root turnover.
Minirhizotrons Use for Root Studies
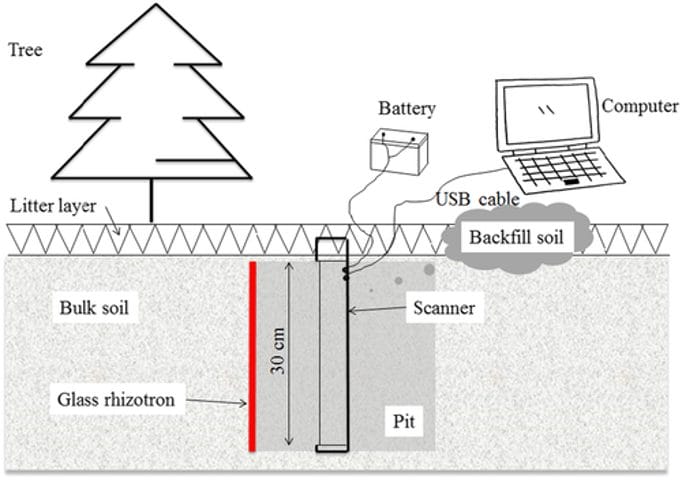
Figure 1: “Schematic diagram of root scanning system. Rhizotrons were vertically installed in a larch plantation in northeastern China. The rhizotron pits were immediately backfilled after finished scanning” Huo and Cheng, 2019. (Image credits: https://doi.org/10.1002/ecs2.2793)
Subscribe to receive our monthly round-up of articles.
Scientists increasingly use minirhizotrons to study root structure, production, longevity, mycorrhizal associations, and disease attacks. They collect repeated images or videos to follow the fate of individual roots over time.
Plants allocate fixed carbon resources to roots for absorption, water, and nutrients. After death, root decomposition adds carbon and other nutrients to the soil. Root turnover rates measure how often plants replace their roots and add organic matter to the soils.
Soil processes and climate influence root turnover, so understanding global warming-driven changes is essential. Root turnover rates need to be studied for their impact on individual plants and their contribution to various ecosystem processes. Scientists are also trying to understand differences in root turnover rates across plant types and ecosystems to find which plant groups or regions are more sensitive to climate change.
Tracking the measurement of root turnover is particularly challenging as the growth and death of roots occur simultaneously. Therefore, scientists are trying minirhizotrons in managed and natural ecosystems, as the technique has successfully measured several other root parameters and processes.
Minirhizotron Use
The essential steps to remember when using a minirhizotron for root turnover measurements are tube installation, number of image captures, and image analysis.
Root Tube Installation
Root tubes are usually around 0.5 to 2 meters long and come with water-tight caps. Their installation is a critical step. The tube materials used are transparent glass, acrylic, or polycarbonate. The materials do not influence root growth.
The angle of tube insertion can be critical. Tilted tubes are preferable to vertical or horizontal tube placement. Horizontal tubes, though cheaper to install, as no deep digging is necessary, cause considerable soil disturbance.
Good soil-to-tube contact is essential without soil compaction. The soil around the tube must have the same conditions as the rest.
Any tube installation causes disturbance and can generate artificial root growth patterns. Therefore, 6-12 months after tube installation is ideal for roots to recolonize the disturbed areas around the tubes and for soil processes to return to pre-disturbance levels. To study root turnover, tube movement must be prevented by securing the tubes to allow for repeated image collection.
Frequency of Data Collection
Image scan frequency depends on the root parameters studied in time series experiments.
Data collection is expensive and requires time. However, choosing adequate samples is crucial to all experiments.
To study root turnover, an interval of eight weeks between scans is too long, as fine roots can be produced and die even within this time. So, root turnover rates will be underestimated. Some recommend a month. Another suggestion is to collect data once in two weeks at depths less than 12 cm; scans at deeper depths can have a longer interval.
Image Capture and Analysis
The cameras used for root image capture have high resolutions varying from 100 to 1200 DPI and can take digital color images or videos. The thin camera system is inserted into the root tubes to take scans at the required depth using precise positioning systems. The camera head can be rotated 360 degrees to scan all the soil around the root tubes.
The same camera is used with several root tubes and as many times as necessary. The data collected is non-destructive and requires only seconds, making it useful for time series experiments like root turnover studies. Recording quantitive data in volumetric units like aboveground plant measures is helpful for root production and turnover studies.
Earlier, manual or semi-automatic analysis of the scans and images was necessary. The work was tedious, and differentiating roots from their surrounding soil was often challenging, especially dark ones. Developing appropriate software for root image analysis has been the focus of considerable research and technology development.
Algorithms can help in root detection and measurement and cut analysis time. However, in many cases, discrepancies remain between minirhizotron estimations and those of reference core sampling and carbon isotope methods.
Analyzing fewer frames without compromising efficiency to reduce sampling efforts is possible. For example, Dubach & Russelle (1995) analyzed every second image of alfalfa and birdsfoot trefoil roots from both sides of the tube without losing accuracy. However, precision was lost when images from only one side or consecutive images were used.
CID Bio-Science Inc. offers two root imagers, the CI-600 Insitu Root Imager, and the CI-602 Narrow Gauge Root Imager. The company also offers software in a package called RootSnap. It differentiates between root color and soil and maps roots in less than six seconds, aiding in the measurement of root turnover.

Calculating Root Turnover
There are various approaches to measuring root turnover.
Using the minirhizotron, root turnover can be calculated using the annual root length production ratio to average live root length. The second method is the inverse of median root longevity, or when 50 percent of roots are dead.
According to Gill and Jackson (2000), another method calculates the annual production ratio to the maximum standing crop.
Root turnover = annual belowground production/ maximum belowground standing crop
Minirhizotrons are better than other methods for measuring root turnover rates. They help estimate large and fine root turnover, while other methods, like the radiocarbon technique, are better for larger roots and large, finer shoots only. Meanwhile, using discrete samples, soil and ingrowth cores estimate standing biomass and relative growth.
These varying methods make comparison of turnover rates difficult.
The widespread adoption of minirhizotrons for turnover studies is still limited because automatic image analysis methods are absent. Some scientists are working on providing models using neural networks that will offer fine-grained analysis of root growth and decay comparing two images. The model will recognize the visible roots in both pictures to determine turnover.
What We Know of Root Turnover
Some research findings based on root images from minirhizotrons are as follows:
- Roots of different branching orders have varying turnover times in Larix gmelinii Turnover rates increased with branching orders. Turnover time for first-order roots is 284 days; for second-order roots, it is 994 days. Third to fifth-order branch roots survived over four years (the experiment duration).
- Global patterns of root turnover were revealed by comparing ecosystems and plant functional types by reviewing 190 studies.
- Root turnover rates increased with mean annual temperature in total shrub biomass and fine roots in grasslands and forests. No global relationship was found between precipitation and root turnover.
- Plant functional groups have varying turnover rates. Turnover rates were lowest for entire tree root systems at 10 percent, followed by shrub total roots at 34 percent. Fine-roots turnover was 53 percent in grasslands, 55 percent in wetlands, and 56 percent in forests.
Better Root Turnover Understanding
Minirhizotons have enhanced our understanding of root turnovers by considering factors like branching orders. As more studies use this easy in-situ technique for a complex process like root turnover, a picture of climate change’s impact on root turnover is emerging.
Sources
Ball-Coelho, B., Sampaio, E. V. S. B., Tiessen, H., & Stewart, J. W. B. (1992). Root dynamics in plant and ratoon crops of sugar cane. Plant and soil, 142, 297-305.
Cheng, W., Coleman, D. C., & Box Jr, J. E. (1990). Root dynamics, production and distribution in agroecosystems on the Georgia Piedmont using minirhizotrons. Journal of Applied Ecology, 592-604.
Dubach, M., & Russelle, M. P. (1995). Reducing the cost of estimating root turnover with horizontally installed minirhizotrons. Agronomy Journal, 87(2), 258-263.
Hendrick, R.L., & Pregitzer, K.S. (1996). Applications of minirhizotrons to understand root function in forests and other natural ecosystems. Plant Soil 185, 293–304. https://doi.org/10.1007/BF02257535
Huo, C., & Cheng, W. (2019). Improved root turnover assessment using field scanning rhizotrons with branch order analysis. Ecosphere, 10(8), e02793.
Gill, R.A., & Jackson, R.B. (2000). Global patterns of root turnover for terrestrial ecosystems. New Phytologist 147, 13-31. doi:10.1046/j.1469-8137.2000.00681.x
Pregitzer, K. S., Zak, D. R., Loya, W. M., Karberg, N. J., King, J. S., & Burton, A. J. (2007). The contribution of root–rhizosphere interactions to biogeochemical cycles in a changing world. In The Rhizosphere (pp. 155-178). Academic Press.
Vamerali, T., Bandiera, M., & Mosca, G. (2012). Minirhizotrons in modern root studies. Measuring roots: an updated approach, 341-361.
Gillert, A., Peters, B., von Lukas, U.F., Kreyling, J., & Blume-Werry, G. (2023). “Tracking Growth and Decay of Plant Roots in Minirhizotron Images,” 2023 IEEE/CVF Winter Conference on Applications of Computer Vision (WACV), Waikoloa, HI, USA, 2023, pp. 3688-3697, doi: 10.1109/WACV56688.2023.00369.
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science