June 14, 2022 at 8:27 am | Updated July 1, 2022 at 10:26 pm | 7 min read
- Understanding underground carbon fluxes in wetlands has been challenging since the standard methods have primarily been destructive.
- Non-destructive minirhizotrons were tested as a possible solution to studying underground dynamics.
- Minirhizotron installation, anchorage, image capture and analysis, and data upscaling recommendations are made to meet the needs of the unique environment of wetlands.
A Methodological Framework for Using Minirhizotrons
To understand soil carbon fluxes, we have to know about root dynamics. The standard methods to study wetland roots are based on sequential coring, which is destructive, laborious, and tedious. Large rhizotrons observatories are expensive. Minirhizotrons allow non-destructive and repeated data collection that is also cost-effective and could be the solution to studying wetland roots.
Minirhizotrons have been used often in uplands, but the common perception is that they are not suitable for wetlands. Therefore, until recently there have been very few research projects using minirhizotrons in this environment.
Iversen, Murphy, Allen, Childs, Eissenstat, Lilleskov, Sarjala, Sloan, and Sullivan, a team of environmental and biological scientists, wanted to develop a methodological framework to use minirhizotrons to study fine and total roots in wetlands.
Subscribe to receive our monthly round-up of articles.
They decided to focus on wetland ecosystems that shared two characteristics, poor drainage, and a deep organic soil horizon. Several wetland habitats have these properties, including peatlands, marches, swamps, boreal forests, and arctic tundra. These represent ecosystems with a range of plant habits from predominant mosses in bogs, graminoids in rich fens, rainforest trees in tropical swamps, and black spruce and tamarack in the boreal forest. Some of these ecosystems have mycorrhizae that are important in nutrient cycling.
As part of the project, the scientists aimed to suggest methods of installation and use suitable for all these wetland conditions. Their recommendations are described below. Some suggestions are uniquely for wetlands, while others are also relevant for other ecosystems.
Minirhizotron Installation
Installing the root tube in wetlands is not as easy as in uplands, and the scientists had to find different solutions for a range of challenges:
- To get past the thick root density in the upper soil layer in peatlands, the scientists suggest making pilot holes to core through for the upper 20cm of peat. The root tubes can then be pounded through this part to the lower, less dense peat. The scientists suggest that care should be taken not to create a large hole at the surface to prevent gaps and light leaks.
- Another possibility is to use a machined cone or point fitted to the tip of the minirhizotron to help in installation, to prevent applying pressure at the bottom, or increase peat density during installation.
- To limit disturbance to the ecosystem and facilitate future scanning, scientists suggest adding a boardwalk.
- The installation method could also influence the material of the root tube. Due to potential effects on root growth, acrylic tubes are better than cellulose acetate butyrate (CAB). However, both these materials are better than others, like glass and polycarbonate.
- Minirhizotrons should be sealed at the bottom with adhesive and non-toxic marine-grade silicone to deal with flooding in areas with a shallow water table. The tubes should be cleaned before inserting the camera for scanning to deal with leakage.
- Water percolation from the surface into the root tube should be prevented using soil sleeves, rubber grippers and stoppers, PVC endcaps, and tape.
- Vertical installations are difficult, and an angle of 45 degrees is best and is achieved with the help of a guide and protractor.
Properly installing root tubes will also facilitate studying microtopography in wetlands, which can be crucial as it can form nutrient gradients.
The scientists recommend using several minirhizotrons in a plot as they are not expensive. It would help to study the spatial heterogeneity of root distributions and protect against extreme weather. Some ecosystems need more root tubes, like those with graminoids, which have roots deeper than trees; with depth, the roots become sparse, and more minirhizotrons are required.
Minirhizotron Anchorage
Long-term stability of minirhizotrons is an issue because of winter frost-heave and fluctuating water levels. The low bulk density of organic matter also makes soils less firm in wetlands. Therefore, all wetland studies use extra anchorage to prevent rotation or vertical movement of the root tubes.
Previous research used anchors made of wood, metal, or steel. They were installed at a depth of one to three meters and secured the minirhizotrons. In the current study, the scientists decided it was also necessary to consider the acidic and water-logged conditions and the possibility of redox dynamics. Therefore, they recommend Teflon-coated stainless steel rods or fiberglass to prevent metal contamination of soils.
Another option is to install the minirhizotrons deep into the soil for stability, even if deep soils are not examined. Anchorage used in upland studies can also be used in wetlands.
To avoid dislocating or displacing the minirhizotrons, the index handle can be foregone to prevent the process of detaching and attaching the index handle cuff to the root tube. Instead, the root tubes are etched or marked with rectangles.
Root Image Capture and Analysis
Image capture of roots in minirhizotrons has come a long way since 1937, when a mirror combined with an electric bulb apparatus was the first technique used.
Currently, root scans are taken by color digital camera, video and high-resolution scan. Three prominent brands offer image capture systems, including CID Bio-Science Inc. The company provides two Root Imagers, the CI-600 In-Situ Root Imager for minirhizotrons of an inner diameter of 6.35 cm. The CI-602 Narrow Gauge Root Imager is for root tubes that are 5 cm wide.
Both root imagers make high-resolution scans in as little as 30 seconds, with higher scanning resolutions requiring more time. The camera head can be rotated 360 degrees to scan the entire tube, recording pictures in 20 cm increments.
Root Image Analysis
There are several software options on the market that analyze root images. The CID Bio-Science Inc. provides the RootSnap! Software, preloaded on the tablet that comes with the device, to quickly differentiate roots from the soil and calculate root dimensions like length, width, area, volume, and branching angles for individual and total roots.
In the wetlands, keeping track of roots over time can be a problem even with minirhizotrons because roots are lost from image frames for reasons other than mortality. These reasons could be fluctuating water table, frost-heave, and shifting or subsidence of peat. The slow decay can also complicate the calculation of the lifespan of roots, as differentiating between dead and live roots can become difficult.
Additionally, species-based differences in root growth, branching, and appearance are all factors that scientists must keep in mind to calculate root turnover.
Frequency of Root Imaging
The frequency of image capture depends on the scientific question.
Images can be taken for a few hours or days to record root phenology or short-term responses, done a few times a year.
Studying root production and mortality will require more frequent data collection. In wetland studies, timing can differ, ranging from every 1 – 2 weeks up to monthly scanning events. Image capture after three days is recommended to limit underestimating root production in uplands, and scientists point out that root turnover could occur at the same rate in wetlands. Since little is known about root dynamics, image capture is also needed in winter to estimate annual production.
Following minirhizotron tube installation in established root systems, it is important to take environmental stabilization into account. Depending on the root system, it can take six months or more for root growth to stabilize to levels similar to undisturbed soils. For root turnover, scientists may have to wait four years to sample standard growth patterns. Long-term studies are needed to collect this information and recommend optimal data collection intervals.
Scaling Root System Data and Integration in Models
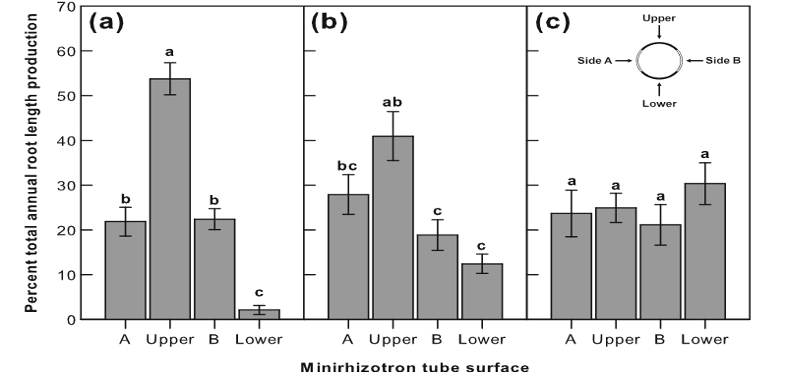
To explore the contribution of fine roots to ecosystems, minirhizotron image scaling is necessary. Upscaling is completed in two steps, from the minirhizotron surface to soil volume and from root length and width to root biomass. Once complete, this can all provide vital information on ecosystem carbon and nutrient recycling, see Figure 1.
Models based on root dynamics must resolve issues related to water table depth, root growth and turnover, root physiology changes with depth, changes in rooting outside the growing stage, as well as differences in root allocation and depth based on plant types. However, despite existing limitations, minirhizotrons remain an excellent method to explore current research questions regarding fine roots and recent advances in the technology are looking to make this even easier.
Fine Roots in Wetlands
Wetland soil carbon is crucial in carbon dioxide and methane fluxes. Underground wetland carbon deposits are vital as they store more than a quarter of terrestrial soil carbon. Carbon accumulates in wetlands because the decomposition rate is slower than biomass production rates due to acidic and oxygen-poor soil environments. As a result, nutrient recycling is poor, and only a few species adapted to the conditions of the wetlands grow here.
The wetland species’ fine roots are aerenchyma and can penetrate and aerate waterlogged soils. Fine roots are responsible for water and nutrient uptake and play an essential role in nutrient recycling in the ecosystem. The high turnover of fine roots also accumulates more organic matter adding to soil carbon. Despite their importance to the wetlands for the living plants and carbon stock, the long-term dynamics of fine roots have not been examined.
Technology
A thorough study of little-known phenomena like underground dynamics of wetlands is advanced by the availability of new non-destructive and efficient technology. Taking repeated images of the same soil zone and depth is crucial to learning the basics about root systems in challenging ecosystems like wetlands. More basic information on species-specific rooting and depth distribution will help scientists explore more complex questions on dynamics.
—
Vijayalaxmi Kinhal
Science Writer, CID Bio-Science
Ph.D. Ecology and Environmental Science, B.Sc Agriculture
Read the original peer-reviewed paper to find out more:
Iversen, C. M., Murphy, M. T., Allen, M. F., Childs, J., Eissenstat, D. M., Lilleskov, E. A., Sarjala, T. M., Sloan, V. L., & Sullivan, P. F. (2011). Advancing the use of minirhizotrons in wetlands. Plant and Soil, 352(1-2), 23–39. https://doi.org/10.1007/s11104-011-0953-1
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






