March 2, 2026 at 7:37 pm | Updated March 2, 2026 at 7:37 pm | 6 min read
- Chlorophyll fluorescence (ChF) can detect individual and multiple nutrient deficiencies simultaneously.
- The method involves non-destructive, precise, real-time measurements.
- A species-specific approach is necessary when developing ChF-based technology to detect multiple nutrient deficiencies.
The main aim of agricultural practices is to optimize conditions and resources essential to maintaining productivity. Instead of determining the levels of a single nutrient, it may be better to assess the nutrient status of multiple elements, as this more accurately reflects growing conditions. Most methods for estimating plant nutrient status are slow and destructive. In this article, we discuss chlorophyll fluorescence, which is emerging as a non-destructive, reliable, and precise method for estimating overall crop nutrient status to advise sustainable fertilizer applications.
Sustainable and Precision Agriculture
Estimating nutrient status in the field in real time is essential to provide economic and ecological benefits. People are moving away from generalized fertilizer recommendations because they lead to excessive use of chemicals or manure, which pollutes soil and water. To determine optimal nutrient doses for crops, it is not enough to assess soil fertility and nutrient availability. It is also necessary to find the exact physiological status of the plants. Determining the nutrient status of plants in real time allows growers to apply nutrients at the right time and in the necessary amounts.
Usually, physical examination of symptoms or laboratory chemical analysis is used to detect nutrient deficiency. However, by the time visual symptoms appear, some of the damage from nutrient deficiency is already irreversible, and a reduction in crop yield quantity and quality can be expected. Hence, remote and nondestructive methods for detecting nutrient deficiencies are increasingly in demand.
Subscribe to the CID Bio-Science Weekly article series.
By submitting this form, you are consenting to receive marketing emails from: . You can revoke your consent to receive emails at any time by using the SafeUnsubscribe® link, found at the bottom of every email. Emails are serviced by Constant Contact
Precision agriculture aims to detect nutrient deficiencies before visual symptoms appear and to take timely, proactive measures, while accounting for spatial heterogeneity in crop performance. Detecting nutrient deficiencies in a timely manner not only enhances crop productivity but also makes food production more sustainable.
Most efforts, even in precision agriculture, so far have focused only on estimating nitrogen (N) deficiencies, the most important nutrient, which is also lacking in many soils worldwide. The inability to detect phosphorus and potassium deficiencies remotely is considered a drawback of precision agriculture. Hence, recommendations for macronutrients phosphorus (P) and potassium (K) lag behind those for nitrogen (N) and can affect plant health and productivity.
Moreover, plants require many other nutrients. Besides temperature, precipitation, and water, the needs for macro- and micronutrients shape a crop species’ niche. The requirements of each crop species vary because they occupy different ecological niches. Plant nutrient status determination for crop species and cultivars should therefore provide information on the optimal range for combinations of multiple nutrients, not just for N or other individual elements, to reflect real-life situations.
Chlorophyll fluorescence (ChF) is currently used to identify nitrogen deficiencies, and research suggests it can be extended to detect multiple nutrients simultaneously.
Chlorophyll Fluorescence
Sunlight striking leaves is absorbed by chlorophyll a; part of it is used in photosynthesis, part is emitted as heat, and the rest is emitted as light, called chlorophyll fluorescence. Chlorophyll a is present in photosystems I and II (PS I and PS II); however, only the portion present in PSII is actively involved in photosynthesis. Light absorbed by PSII is used to split water into oxygen and electrons. Electrons are transferred by the electron transport system (ETR) through PSII to PSI to reduce NADP+ (Nicotinamide adenine dinucleotide phosphate), and the remaining electrons are finally accepted by carbon dioxide in a process called photochemical quenching. Heat loss occurs through a process called non-photochemical quenching (NPQ). Chlorophyll fluorescence is the light remaining after these two processes.
Chlorophyll fluorescence can detect stress in plants, including nutrient deficiency. For example, when plants lack nitrogen, chlorophyll levels decrease, reducing photosynthesis. As a result, a greater proportion of incident sunlight is lost as chlorophyll fluorescence.
There are several fluorescence parameters that can be used for nutrient deficiency detection:
- The Fv/Fm ratio measures the maximum quantum yield efficiency of photosystems or photosynthetic rate. Any factor that reduces photosynthesis will lower the ratio and indicate stress. However, this ratio is non-specific and often not sensitive.
- Other fluorescence parameters, such as NPQ and ETR, can also be used to estimate nutrient deficiency.
These parameters can be obtained from light-adapted leaves using the saturation pulse method.
OJIP transients
Another method being researched is the use of chlorophyll a fluorescence OJIP transients from dark-adapted leaves during a short actinic light pulse in a fluorimeter. The fluorescence rise from the initial (Fo) to the maximum (Fm) phase is recorded through the O, K, J, I, and P stages. The fluorescence curve rises from the Fo to Fm stages, and this polyphasic transient is described by a mathematical model called the OJIP test. It provides insights into the photochemical efficiency of the photosynthetic apparatus, the structural and functional attributes of PS I and PS II, particularly electron transport, and quantum yields. The OJIP method was developed in 1995 by Strasserf et al.
The ChF transients are considered “fingerprints” that can be used to assess different types of stress. It can also differentiate among stress responses based on varying stress intensity, the plant developmental stage at which stress occurs, and genotypic variation.
The OJIP test has been used in research since the turn of the century to detect nutrient deficiencies.
Detection of Nutrient Deficiency
Chlorophyll fluorescence signals from non-destructive OJIP have been used to detect individual nutrient deficiencies of the macronutrients nitrogen (N), phosphorus (P), and potassium (K) in many species. Chlorophyll fluorescence transients can also detect combinations of nutrient deficiencies occurring simultaneously, such as NK, KP, NP, and NPK. Detection was possible even before the visual symptoms of the combined deficiencies developed in two crops, wheat and maize.
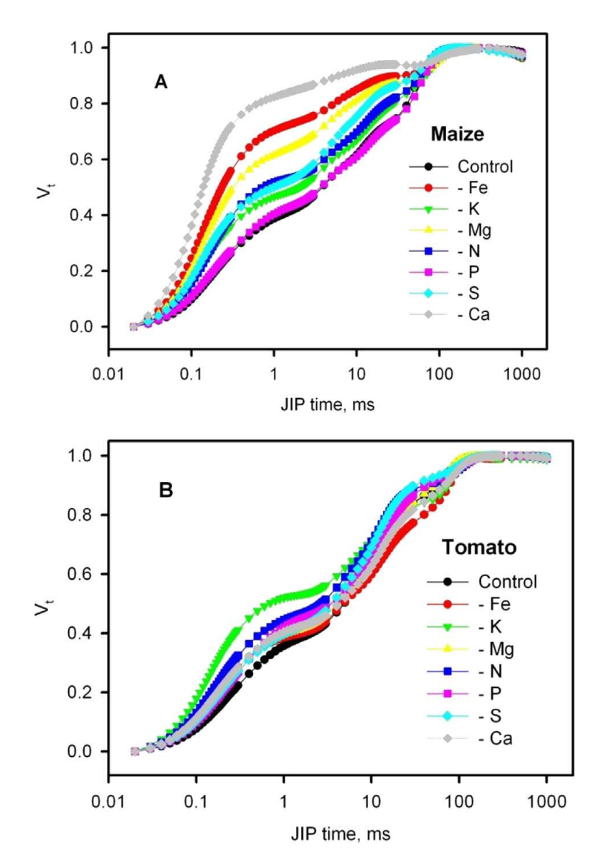
Figure 1: “Effect of nutrient deficiencies on relative variable fluorescence at the J-step Vt [Vt=(Ft-Fo)/(FM-Fo)], Kalaji, et al. (2014). (Image credits: http://dx.doi.org/10.1016/j.plaphy.2014.03.029)
In addition, deficiencies of several other nutrients necessary for plant growth, development, and physiological functioning, including secondary macronutrients calcium (Ca), magnesium (Mg), and sulfur (S), and micronutrients manganese (Mn), copper (Cu), zinc (Zn), and iron (Fe) have also been detected in OJIP studies.
The absence of these nutrients can affect the structure and function of the photosynthetic apparatus, and the resultant changes in chlorophyll fluorescence are detectable in OJIP transients. The different ways in which the nutrients change photosynthesis are given below:
- Deficiency of nitrogen, magnesium, iron, and zinc directly affects chlorophyll synthesis, the site of photosynthesis.
- Nitrogen, sulfur, and iron deficiencies directly affect the biosynthesis of proteins involved in photosynthetic reactions.
- Phosphorus deficiency influences photosynthesis by slowing the production of ATP (Adenosine Triphosphate) and NADP involved in the Calvin cycle, which can affect the parameters ETR, QP, and ϕPSII (Quantum yield of photosystem II).
- Calcium deficiency affects membrane stability and reduces PSII activity.
- Calcium and potassium deficiency affect osmotic homeostasis and cell signaling during stress response.
- Calcium and magnesium deficiency lead to a significant decrease in electron donation by the oxygen-evolving complex.
- Sulphur deficiency limits electron transport by reducing PS I activity.
Deficiencies in other nutrients cause reduced growth and biomass accumulation, as well as water imbalance. Lower biomass reduces photosynthesis because there is less demand for assimilates. This low sink is another way phosphate deficiency affects photosynthesis.
An experiment by Kalaji, et al. (2014), showed that N, P, K, Mg, S, Ca, and Fe deficiency in tomato and maize affected the ChF curves. In nutrient-deficient plants, there is a reduction in photochemical efficiency, an increase in non-photochemical (heat) dissipation, and a decrease in the number of active PSII reaction centers. However, individual nutrient deficiencies have specific responses in limiting PSII and PS I activity.
Moreover, responses to nutrient deficiency occurred at different parts of the photosynthetic apparatus in tomatoes and maize, indicating interspecific differences in ChF responses; see Figure 1. Therefore, a species-specific approach is necessary when developing ChF-based technology to detect multiple nutrient deficiencies in precision agriculture.
Tools for Measuring Chlorophyll Fluorescence
The quick OJIP transients are determined by fluorimeters. However, it is possible to acquire essential fluorescence parameters like Fv/Fm, NPQ, and ETR from other tools such as the
CI-510CF Chlorophyll Fluorescence Module, which is used with the CI-340 Handheld Photosynthesis System. The tool can be used in vivo for nondestructive, real-time estimation of chlorophyll fluorescence parameters in nutrient-deficiency research or precision agriculture.
Contact us at CID BioScience Inc. to learn more about the CI-510CF Chlorophyll Fluorescence Module for nutrient deficiency detection.
Sources
Jaszczuk, Z. M., & Bąba, W. (2023). Detection of multi-nutrients deficiency in cereal plants by the use of chlorophyll fluorescence. Journal of Water and Land Development, 224-233. DOI:10.24425/jwld.2023.148447
Gupta, R., Sharma, R.D., Rao, Y. R., Siddiqui,……& Tuteja, N. (2021). Acclimation potential of
Noni (Morinda citrifolia L.) plant to temperature stress is mediated through photosynthetic electron transport rate, Plant Signaling & Behavior, 16:3, 1865687, DOI: 10.1080/15592324.2020.1865687
Kalaji, H. M., Oukarroum, A., Alexandrov, V., Kouzmanova, M., Brestic, M., Zivcak, M., … & Goltsev, V. (2014). Identification of nutrient deficiency in maize and tomato plants by in vivo chlorophyll a fluorescence measurements. Plant physiology and biochemistry, 81, 16-25. https://doi.org/10.1016/j.plaphy.2014.03.029
Kalaji, H. M., Bąba, W., Gediga, K., Goltsev, V., Samborska, I. A., Cetner, M. D., Dimitrova, S., Piszcz, U., Bielecki, K., Karmowska, K., Dankov, K., & Kompała-Bąba, A. (2018). Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynthesis research, 136(3), 329–343. https://doi.org/10.1007/s11120-017-0467-7
Kanash, E. V., Sinyavina, N. G., Rusakov, D. V., Egorova, K. V., Panova, G. G., & Chesnokov, Y. V. (2023). Morpho-Physiological, Chlorophyll Fluorescence, and Diffuse Reflectance Spectra Characteristics of Lettuce under the Main Macronutrient Deficiency. Horticulturae, 9(11), 1185. https://doi.org/10.3390/horticulturae9111185
Liu, Y., Gong, C., Pei, W., Fan, K., & Shen, W. (2024). Chlorophyll a fluorescence as a tool to monitor physiological status in the leaves of Artemisia ordosica under root cutting conditions. Frontiers in Plant Science, 14, 1308209.
Ripoll, J., Bertin, N., Bidel, L.P.R., & Urban, L. (2016) “A user’s view of the parameters derived from the induction curves of maximal chlorophyll a fluorescence: Perspectives for analyzing stress,” Frontiers in Plant Science, 7, 1679. https://doi.org/10.3389/fpls.2016.01679.
Stirbet, A. (2011). On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and photosystem II: basics and applications of the OJIP fluorescence transient. Journal of Photochemistry and Photobiology B: Biology, 104(1-2), 236-257.
Strasserf, R. J., Srivastava, A., & Govindjee. (1995). Polyphasic chlorophyll a fluorescence transient in plants and cyanobacteria. Photochemistry and photobiology, 61(1), 32-42.
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






