March 16, 2026 at 4:50 pm | Updated March 16, 2026 at 4:50 pm | 11 min read
- Root traits in plants exhibit spatial and temporal adaptations to survive in the unique combination of conditions found in various natural ecosystems.
- Root system’s architecture, rooting depth, biomass, and root-to-shoot ratio are the traits that have been most often monitored across ecosystems.
- Root systems differ in their responses to nutrient and water availability and to various abiotic stresses that can be specific to an ecosystem.
Plants develop traits in response to their environment to increase their chances of survival and reproduction. The diversity of above-ground vegetative traits, which are often distinct across different habitats and ecosystems, is well studied, not least because they are visible. Knowledge of ecosystem-specific diversity in root trait structures has gradually increased over the past two decades, aided by modern minirhizotron technology. This article summarizes the major differences in root morphology observed so far across ecosystems.
Root Plasticity
The root system’s architecture and other morphological traits, developed in response to the environment, determine the ability to explore the soil for nutrients and water, provide structural support, facilitate coexistence, and adapt to stress. Phenotypic plasticity exists both within and between species, enabling plants to adjust to specific conditions. The diversity in roots is linked to genetic and hormonal control and is interesting as a set of traits that could be replicated or leveraged in plant breeding for crops, or simply to understand how natural communities function, providing insights into ecosystem dynamics in response to land use changes and climate change.
Plant species rely on plasticity to adapt to their external environment by changing root morphology, architecture, anatomy, and microbial associations. For example, first-order roots in woody plants have the highest diameter in tropical and stable ecosystems, but decrease in deserts and seasonal habitats, which use carbon more efficiently by growing thinner roots to colonize new habitats.
Subscribe to the CID Bio-Science Weekly article series.
By submitting this form, you are consenting to receive marketing emails from: . You can revoke your consent to receive emails at any time by using the SafeUnsubscribe® link, found at the bottom of every email. Emails are serviced by Constant Contact
Such information on root traits across species, environments, and ecosystems can be crucial for improving predictions on carbon and water fluxes.
Root Distribution
A meta-analysis in 1996 by Jackson et al. of 250 root studies examined how rooting varied across 11 terrestrial biomes to understand and predict ecosystem functioning of carbon, nutrient, and water fluxes. The traits studied were root depth, biomass, and root-to-shoot ratio.
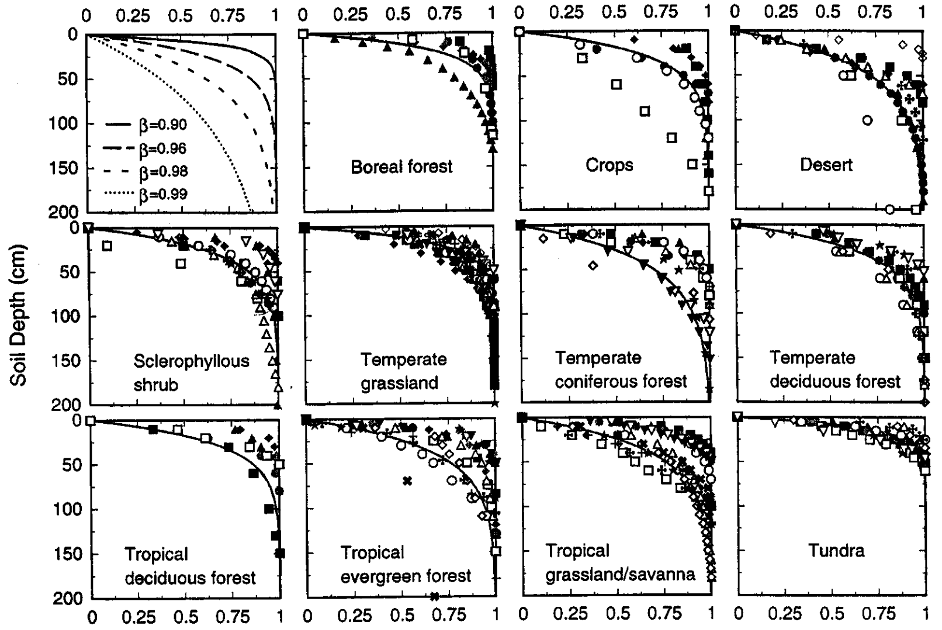
Figure 1: “Cumulative root distribution as a function of soil depth for eleven terrestrial biomes and for the theoretical model of Gale and Grigal (1987) with higher values indicating greater depth.” The symbols represent the studies from which the data were sourced (For more details, see Table 2 in the publication), Jackson et al. (1996). (Image credits: https://www.ehleringer.net/uploads/3/1/8/3/31835701/206.pdf)
Rooting Depth
Global patterns show that 30% of root distribution occurs in the top 10 cm, 50% in the top 20 cm, and 75% in the top 40 cm. However, deeper soils can also be crucial as water and nutrient reserves, especially during drought. For example, species like Eucalyptus and Acacia that grow in drought-prone areas have deep roots. A substantial number of fine roots can be found at depths of around 6.5 meters in Amazonia, Brazil.
Jackson et al. (1996) used the depth coefficient β (Gale & Grigal, 1987) to examine root distribution depth and found the following:
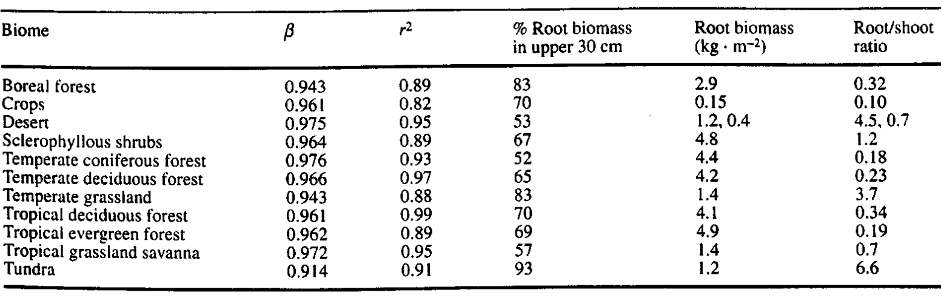
- Globally, around 80-90% of roots are present in the top 30 cm. Ecosystem-specific variations exist in this soil depth, with the most shallow roots in tundra (93%), temperate grasslands (83%), and boreal forests (83%). Tundra has 60% of its roots in the top 10 cm. Root systems are shallow in high latitudes due to permafrost in tundra and boreal forests. In temperate regions, waterlogging is responsible for the shallow root systems.
- Deserts and temperate coniferous forests have the deepest roots, with only 50% in the top 30 cm. Deserts have only 20% of roots in the top 10 cm of soil and grow deeper to avoid the high surface temperatures. Tropical savannas and grasslands also have deeper roots than temperate grasslands.
A pattern also exists for root distributions for plant types across biomes:
- In the top 10 cm of soil, grasses had 44% of roots, shrubs had only 21% of roots, and temperate and tropical trees had 26% of their roots.
- Temperate and tropical trees had 60% of their roots in the top 30 cm of the soil.
Rooting depth for water uptake varies in the ecosystems:
- It is 1.0 meters for croplands, grasslands, and tundra.
- It is 2.0 meters for forests.
Root Total Biomass
The study also tracked total root biomass across biomes and found it differed from patterns seen for rooting depth. Standing root total biomass varies from 0.2 to 5 kg m-2 among the biomes. Large-scale patterns are seen in root biomass occurrence, as shown in Figure 1:
- The least biomass (<1.5 kg. m-2) is in croplands, deserts, grasslands, and tundra. However, cold deserts had three times as much root biomass as warm deserts.
- Higher biomass (2-5 kg. m-2) was found in forests and sclerophyllous shrublands. Tropical evergreen forests had the highest root biomass of 5 kg m-2.
Root to Shoot Ratio
The allocation of carbon to grow root and shoot biomass is an adaptive plant strategy.
Jackson et al. found that root-to-shoot ratios (RSR) are highest in tundra (6), followed by cold deserts (4.5-7) and temperate grasslands (3.7). The greatest variation was found in sclerophyllous shrublands (0.3 to 5), and the ratio was most consistent in forests (0.18 to 0.34), as shown in Table 1.
Qi et al. (2019) also found interesting differences in RSR in plant types across biomes:
- Trees had a lower RSR than grasses and shrubs.
- Angiosperms had a higher average root-to-shoot ratio than gymnosperms.
- Deciduous species had a higher RSR than evergreen plants.
- RSR is negatively correlated with plant height and shoot biomass.
Root-to-shoot ratios are sensitive to environmental conditions; RSR is positively correlated with latitude and elevation but negatively correlated with precipitation and mean annual temperature. Overall, it suggests that plants adapt to environments and adjust their growth strategies by allocating more photosynthates to roots in stressful, low-nutrient, and adverse climates. Therefore, the highest RSRs are found in the tundras as indicated by many studies.
Table 1. “Values of β and r2 for the Gale and Grigal model, the percentage of roots in the upper 30 cm, average standing root biomass (kg. m-2), and root: shoot ratios for eleven biomes,” Jackson et al. (1996). (Image credits: https://www.ehleringer.net/uploads/3/1/8/3/31835701/206.pdf)
Drivers for Root Adaptations
The root morphological trends observed across ecosystems can be explained by drivers such as nutrient availability and unavailability, as well as extremes in temperature and precipitation. These can include excesses or deficiencies, such as high or low temperatures, more or less rainfall, waterlogged conditions, or salinity.
Plants need soil mineral nutrients and tolerance to stresses for survival, growth, and productivity. In crops, these stresses can affect yield. Plants have evolved root adaptations specific to their ecosystems’ unique growing conditions, helping them survive and thrive under the stresses they encounter.
In the following sections, we discuss common spatial and temporal root adaptations in architectural and morphological traits across ecosystems, including extreme environments such as deserts, mangroves, aquatic habitats, and nutrient-deficient environments (see Figure 2).
Nutrient Deficient Environments
Nutrient deficiencies are found worldwide, from biodiversity-rich tropical rainforests to cold and warm deserts. The cause of deficiency can vary and depend on climate and geological conditions:
- Rainforests: Nutrient deficiency is caused by heavy rainfall, which leaches nutrients from the topsoil. Weathering of rocks can acidify soils and release nutrients toxic to plants. Interestingly, nutrient deficiency is one of the drivers of high species numbers in rainforests.
- Deserts: Low rainfall and nutrient mobility, and less organic matter and nitrogen production are the causes of nutrient deficiency in desert soils.
- Tundra: Similarly, in cold tundra, the restricted plant growth limits nitrogen cycling, making soils less fertile.
Nutrient deficiency can explain the trends in deep roots observed in global studies of root length. However, other root traits and adaptations are also common.
Root hair production: In response to a deficiency of phosphorus and iron in soil, roots grow longer and denser root hairs to improve uptake of the immobile nutrients, in tomatoes, spinach, rape, and A. thaliana.
Anatomical plasticity: Roots exhibit anatomical plasticity, changing tissue composition in response to nutrient deficiencies, such as phosphorus, nitrogen, and sulfur. Aerenchyma, with low metabolic costs, increases to support root growth for deeper soil exploration in maize and common beans.
Cluster roots: Proteoid or cluster roots are a common adaptation in less fertile soils. The roots develop determinate rootlets with dense root hairs resembling bottlebrushes. Deficiencies of phosphorus and iron trigger the formation of cluster roots in several plant families growing in low-fertility soils, such as Betulaceae, Cucurbitaceae, Cyperaceae, Casuarinaceae, Eleagnaceae, Leguminosae, Proteaceae, Myricaceae, Moraceae, and Restionaceae. Cluster roots are also developed in nutrient-rich soil pockets to increase nutrient mobilization. Cluster root formation occurs alongside increased root exudate secretion in the rhizosphere to recruit beneficial microbes that mobilize nutrients, such as phosphorus.
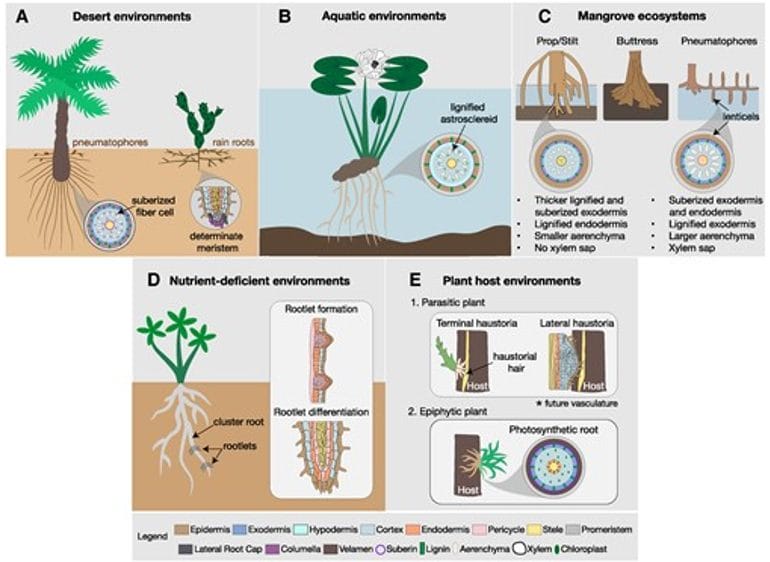
Figure 2. “Root system architecture and anatomical responses to different habitats. A) Date palm tree on the left and cactus plant on the right; B) Water lily root; C) Common root structures of mangrove species; D) White lupin plant with rootlets; E) Parasites and epiphytes’ roots.” Ramachandran et al. (2025). (Image credits: https://academic.oup.com/plphys/article/197/1/kiae586/7917389)
Deserts
Deserts are regions with less than 25 cm of annual rainfall, with spatial and temporal variations in soil water availability. Deserts have shallow-rooted ephemerals and perennials, and deep-rooted perennials. Though root distribution varies by species, some common patterns are observed in desert plants.
- Deep and lateral roots: Desert plants have both deep and lateral root systems to access water at varying soil depths. Growing shallow lateral roots quickly allows seedlings to absorb water and nutrients from the soil surface and survive. In arid regions, cacti of the Cactoideae subfamily develop determinate primary roots that mature within a few days and differentiate into root hairs and lateral hairs for enhanced soil exploration. Plants of the same family growing in regions with moderate rainfall grow indeterminate roots.
- Root contraction: Desert plants from Agavaceae, Asteraceae, and Cactaceae exhibit anatomical changes that result in root contraction, pulling the shoot deeper into the cooler soil and helping tide over high surface temperatures. These roots also exhibit higher hydraulic conductivity, thereby improving water uptake.
- Rain roots: Some species, such as Opuntia and Agave, can rapidly form rain roots in response to soil moisture and shed them when soil water levels diminish. The roots have aerenchyma with large air cavities that prevent water from flowing out of the roots when the soil dries.
- Pneumatophores: Date palms growing in deserts can develop pneumatophores near the soil surface, below or above the soil, to access moisture in the topsoil. These pneumatophores increase water uptake during rainfall.
Aquatic Ecosystems
In aquatic systems, water is abundant, but plants growing in these habitats encounter hypoxic conditions, nutrient deficiencies, and unstable substrate. Plants can be emergent, submerged, floating-leaved, and free-floating. The root systems of each type of aquatic plant are listed below.
- Emergent species: Emergent plant species have roots in the soil and erect stems with photosynthetic organs extending above the water surface, as in bulrushes. The root system is extensive, and they absorb nutrients from the soil. These plants are found on shallow shorelines and are closest to terrestrial plants in their functioning.
- Submerged species: These plants are found in coastal, freshwater, and estuarine environments. Most are rooted in the substrate and spend their entire lifecycle underwater, except during flowering. The root systems are not very extensive, but they help in nutrient absorption.
- Floating-leaved species: These aquatic plants have roots that anchor them in the substrate in shallow water, and broad leaves with entire margins that float on the water surface, connected by long petioles and stems to the roots. Water lilies and lotus are examples of floating-leaved plants.
- Free-floating: Free-floating plants have roots that are not anchored in substrate but float on the water surface. The root systems remain in the water, absorb nutrients, and their weight helps stabilize plants on the water surface. Some free-floating plants have no roots.
Many adaptations seen in aquatic plants are also present in terrestrial plants that are subject to flooding, such as rice, a wetland species. Rice induces suberization, or the formation of an exodermis layer beneath the epidermis in roots, to act as a barrier to water and oxygen loss from the roots. The exodermis helps transport oxygen to root tips, supporting the rapid growth of the root tips. Wheat grows more aerenchyma in roots to improve flooding tolerance.
Mangroves
Mangroves are periodically flooded and are also areas of high salinity, which can lead to toxic ion accumulation, osmotic stress, and nutritional deficiency. Higher concentrations of sodium also reduce water potential and limit water availability. Since salinity affects one-third of agricultural soils, mangrove species, which are halophytes adapted to these extreme conditions,
can provide valuable insights into breeding salinity-tolerant cultivars. Mangroves are found in tropical and subtropical regions and are also valuable for protecting coasts from storms and erosion.
Halophytes have morphological adaptations such as the following to help them:
Prop/stilt roots: Prop and stilt roots are aerial roots that form as outgrowths of branches and trunks and extend into the soil, providing additional support to the trees. The prop and stilt roots can be submerged during flooding and gather sediment and organic matter to build soil to stabilize the substrate.
Buttress roots: Another trait developed for anchorage is buttress roots, which are large, wide, plank-like above-ground structures that form at the base of the tree. Besides anchorage, buttresses also help transport nutrients and water.
Pneumatophores: A characteristic feature of mangroves, pneumatophores, or breathing roots, are found in many mangrove families to transport oxygen to roots. Pneumatophores are roots formed in flooded conditions and have raised pores called lenticels along with large aerenchyma tissue with lacunae for gas exchange.
Plants as Hosts
Some plant interactions include parasitism and epiphytism. Here, plants that do not grow in the soil use other plants as substrates or as a food source.
Parasitism: Some plants, like mistletoe and striga, are above-ground parasites that depend on plants for food and growing space. Parasitic weeds cause annual crop losses of over $2 billion and impact natural diversity, so learning about their roots can help in controlling them. Plant parasites have developed specialized roots called haustoria. Initially, prehaustoria, or host-penetrative roots, form. Then the parasite develops further to form haustoria, which can be terminal or lateral.
- Terminal haustoria develop when the root apical meristem divides and differentiates to form root hairs that enter hosts to form a connection with the vascular tissue.
- Lateral haustoria are also root hairs developed by the parasite after the terminal haustoria are fully formed to secure the attachment to the host and facilitate nutrient uptake. A hyaline body in the center of lateral haustoria acts as the sink for host metabolites.
Epiphytism: Epiphytes grow attached to host plants but do not invade their tissue for food. Epiphytes have specialized photosynthetic roots to absorb water vapor and nutrients from the air. Epiphytism has developed independently in 23 families, most of which are in Orchidaceae. Orchids have aerial roots to anchor to trees without a root cap or root hairs. Some epiphytes start as terrestrial plants, for example, aroid vines. They grow aerial roots, some of which become anchor roots and secure the vine to the trunk, while others grow down and become feeder roots in the soil.
Minirhizotrons for Root Studies
Minirhizotron systems consist of transparent root tubes installed in soil to a depth of over 100 cm, allowing the roots of the subject plants to grow around them for many weeks. The imager with a high-resolution camera is then inserted to scan the roots at the required soil depths. The imagers offered by CID Bio-Science Inc. can be rotated 360 degrees to scan the entire root tube surface. An accompanying software, RootSnap, analyzes images, distinguishes roots from soil, and calculates root traits such as length, width, area, volume, and branching angle. Minirhizotron systems are suitable for single- or time-series data collection and allow scientists to monitor root growth, turnover, dynamics, diseases, and microbial interactions with mycorrhizae. These tools have become standard equipment for root research in natural ecosystems, greenhouses, and fields.
Contact us to find out more about the CI-600 In-Situ Root Imager and CI-602 Narrow Gauge Root Imager for your research needs.
Sources
Alexandre, G. Soazig, G.H., & Laurent, L. (2025, May 1). The root system of plants: from the shadows to the light. Retrieved from https://www.encyclopedie-environnement.org/en/life/root-system-plants/
Dannowski, M., & Block, A. (2005). Fractal geometry and root system structures of heterogeneous plant communities. Plant and Soil, 272(1), 61-76.
Jackson, R. B., Canadell, J., Ehleringer, J. R., Mooney, H. A., Sala, O. E., & Schulze, E. D. (1996). A global analysis of root distributions for terrestrial biomes. Oecologia, 108(3), 389-411.
Hendrick, R. L., & Pregitzer, K. S. (1996). Applications of minirhizotrons to understand root function in forests and other natural ecosystems. Plant and soil, 185(2), 293-304.
Ma, Z., & Chen, H. Y. (2016). Effects of species diversity on fine root productivity in diverse ecosystems: A global meta‐analysis. Global Ecology and Biogeography, 25(11), 1387-1396. DOI: 10.1111/geb.12488
Norby, R. J., & Jackson, R. B. (2000). Root dynamics and global change: seeking an ecosystem perspective. The New Phytologist, 147(1), 3-12.
Qi, Y., Wei, W., Chen, C., & Chen, L. (2019). Plant root-shoot biomass allocation over diverse biomes: A global synthesis. Global Ecology and Conservation, 18, e00606.
Ramachandran, P., Ramirez, A., & Dinneny, J. R. (2025). Rooting for survival: how plants tackle a challenging environment through a diversity of root forms and functions. Plant Physiology, 197(1), kiae586.https://doi.org/10.1093/plphys/kiae586
Wang, R., Yu, G., & He, N. (2021). Root Community Traits: Scaling-Up and Incorporating Roots Into Ecosystem Functional Analyses. Frontiers in plant science, 12, 690235. https://doi.org/10.3389/fpls.2021.690235
Related Products
Most Popular Articles
- Transpiration in Plants: Its Importance and Applications
- Leaf Area – How & Why Measuring Leaf Area…
- How to Analyze Photosynthesis in Plants: Methods and Tools
- Plant Respiration: Its Importance and Applications
- The Forest Canopy: Structure, Roles & Measurement
- Stomatal Conductance: Functions, Measurement, and…
- Forest & Plant Canopy Analysis – Tools…
- Root Respiration: Importance and Applications
- The Importance of Leaf Area Index (LAI) in…
- 50 Best Universities for Plant Science






